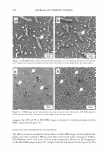
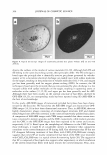
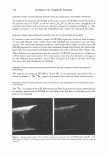
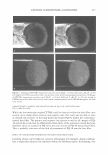
2005 ANNUAL SCIENTIFIC SEMINAR companies. Within those companies, those most directly affected are the Cosmetic Chemists and Microbiologists. The Period After Opening (strictly speaking, not an Expiration Date), is difficult to determine, yet this function of the Chemists and Microbiologists affects the entire company. With rapid product development timelines, this dilemma is intensified and is a challenge for all concerned. The parameters of determining the Period After Opening are many faceted. This requirement is completely new to our industry, and the implementation date was March 11, 2005. Japan has its own set of regulations. They too, have a positive list of preservatives that can be used. If the preservative is not on the positive list, it may not be used to preserve Cosmetic products. Historically, Japan was one of the most difficult markets for preservative acceptance. The .tvlinistry of Health and Welfare (now the Ministry of Health, Labor and Welfare), which is similar to our FDA, had a strict requirement for any ingredient to be approved before being used. For many years, Formaldehyde donor preservatives were not iipproved. Industry provided information citing the safety of these ingredients in other markets. The l\linistry responded by noting their belief that the skin of the Japanese consumer was different and more sensitive than Caucasian skin. Therefore the safetv of wide use in other mnrkets was irrelevant for their safety concerns. Some preservatives frequently used in the U.S. are unacceptable in other countries. They also sometimes have different limitations for use. Unfortunately, it is not until a new market is approached, that these difference� come to light. Foresight and knowledge of the regulations can streamline the development process and can help eliminate additional formulation work. \\'hen dealing with restricted amounts of ingredients, it is absolutely necessary to determine the presence and level of all preservatives in the finished product. Sometimes the presence of preservatives is overlooked, usually due to its presence in ancillary raw materials. l\fany companies have found that there are preservatives present in some raw materials. such a� surfactants and liquid botanical extracts. These preservatives must be taken into account by virtue of their contribution to the finished product. If government authorities test the product off the shelf for preservative levels, they will be checking for all preservatives, regardless of the contributor. Formulators are constantly faced with challenges choosing preservatives that are acceptable in International markets, not just in Japan, and in Europe, but in countries such as Australia. Korea and Taiwan. Recoming knowledgeable in the regulations affecting preservatives in various countries is imperative in designing formulas that will be acceptable in International markets. The definition of an International market must be carefully discussed. Often technical experts consider the U.S , Europe and Japan to be the only International markets. Other countries must also be considered when formulating. Formulators and .Microbiologists alike must now become experts in the field of Regulator�· Affairs in order to stay competitive in our ever shrinking world. 365
366 JOURNAL OF COSMETIC SCIENCE HARMONIZATION OF MICROBIAL TESTING METHODS Joyce F. Graf, Ph.D. The Cosmetic, Toiletry, and Fragrance Association, 110117th Street, NW, Washington, DC 20036 CTFA, through their microbiology committee, has been actively working with many groups to harmonize microbial quality and test methods. These include other organizations such as USP, CO LIPA, /CIA and ISO TC-217 WG1. Recently harmonization on Microbial Limits Guidelines was agreed between the CTFA, COLIPA and /CIA. Manufacturers of personal care products must assure that finished products are free from unacceptable levels and types of microorganisms. From the development of the product and optimizing the preservative system through raw materials testing, manufacturing, and product release testing, the microbiologist is involved. It has been generally accepted that the microbial limit test is a starting point of quality control for member companies however they do not want to hamper the individualities of the cosmetic manufacturers by imposing numerical limits." For the manufacture of personal care products that are also personal care products, the Japanese, European, and United States Pharmacopeias also supply tests. There has been an effort to standardize these methods, although the harmonized preservative effectiveness test differs in many details. Trade organizations such as the Cosmetic, Toiletry & Fragrance Association (CTFA), the European Cosmetic, Toiletry and Perfumery Association (COLIPA), and the Japanese Cosmetic Ingredient Association GCIA) work on behalf of member companies to standardize procedures, disseminate technical information on product use and safety, and interface with governmental/ regulatory agencies to maximize compliance with the minimum amount of enforcement. National and international standard setting organizations have also published preservative efficacy tests. The validity of the AOAC International method, based on the CTFA method, was the subject of a round-robin testing protocol. The ASTM method is under 5-year revalidation, and it is expected to be changed to bring it more into line with the CTFA method. The International Standards Organization (ISO) has developed a number of microbiological methods that will soon be published as ISO international standards. Although the preservative efficacy test has been discussed, there is no formal movement to begin working on this new work area. This presentation will review the existing methods for preservation efficacy, update the efforts at standardizing the CTFA/COLIPA/JCIA microbiological limit guidelines, and review the status of the ISO methods. Microorganisms are opportunistic, and they may grow when nutrients (e.g., foods, drugs, cosmetics, raw materials, product residues, etc.), sufficient water, and environmental conditions are present. Products may provide sufficient nutrients for growth, and preservation is necessary for aqueous products in multiple-use containers. Microorganisms are ubiquitous and capable of adaptation and selection. No method can guarantee microbial control under all conditions. The method for challenge testing involves the inoculation of the product sample with bacteria, yeasts and molds, some of which may either be pathogenic or cause spoilage, followed by testing the inoculated product for viability of the microorganisms at various periods as long as considered necessary. In some cases, these tests are supplemented by chemical tests to measure the amount of preservative in the product. These kinds of chemical tests are required for drug products in the US.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)