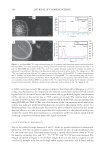
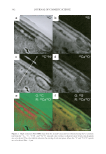
Cu AND Ca UPTAKE IN COLORED HAIR 343 the medulla and in isolated spots in the cortex in both untreated and treated samples. We had some concerns about whether these observations were representative of the extremely low copper concentration in these samples or artefacts of sample preparation. In an attempt to confi rm our ability to reliably map the location of copper ions in these samples, a hair swatch that contained a signifi cantly higher level of copper than would be expected in a consumer situation was prepared. The colorant-treated hair was washed in water containing 1 ppm copper ions for 30 min until the hair was visibly green. ICP-MS analysis confi rmed that the copper level had increased from 120 ppm to 11,000 ppm and that the calcium level had dropped from 8,700 ppm to 4,800 ppm (see Table I). The O− ion beam was used for analysis of positive ions, and the detector was tuned to 40 Ca+ and 63 Cu+. The images obtained are shown in Figure 4. The 40 Ca+ image is similar to what we have seen previously, with the signal concentrated both in the medulla and the cuticle. However, the 63 Cu+signal intensity is still signifi cantly lower than that of 40 Ca+. One pos- sible explanation for this result is that the yield of 63 Cu+ ions from the hair is very much lower than that of 40 Ca+ ions. The relative sensitivity factor, or RSF, for 40 Ca+ in a silicon Figure 3. NanoSIMS line profi les obtained using the Cs+ primary beam on treated hair for 12 C2−, 32 S−, 12 C14N−, and 40 Ca16O− signals. The line scan direction is indicated with an arrow in the 12 C14N image in Fig- ure 2a. Abbr: Ex, exocuticle En, endocuticle. Figure 4. (a) NanoSIMS 40 Ca+ map obtained using the O− primary ion beam from copper-treated Caucasian hair. (b) NanoSIMS 63 Cu+ maps obtained using the O− primary ion beam from copper-treated Caucasian hair. Bar = 10 μm. (c) NanoSIMS line profi les of the 40 Ca+ and 63 Cu+ signal obtained using the O− primary ion beam on copper-treated Caucasian hair. The line scan direction is indicated with an arrow in the 40 Ca+ image in (a).
JOURNAL OF COSMETIC SCIENCE 344 matrix is 1021 to 1022 atoms cm−3, whereas the RSF for 63 Cu+ in a silicon matrix is an order of magnitude lower, 1022 to 1023 atoms cm−3 (22). In this high copper image, the 63 Cu+ signal is observed in the medulla and in isolated spots in the cortex. It is also detected in the cuticle and in a distinct diffusion gradient into the cortex. The diffusion profi le ob- served in the cortex is likely due to the technique used to dose the fi ber. Thus at this stage we can only offer a tentative identifi cation of copper concentrating in the cuticle, in the same way as clearly shown for calcium. CONCLUSIONS The NanoSIMS has proved to be an excellent tool for mapping, with a resolution of up to 50 nm, the location of calcium in both untreated and colored hair. We have demonstrated that the additional uptake of the calcium in colored hair is to a signifi cant extent concen- trated in the cuticle. The technique has also clearly demonstrated the co-location of the calcium with sulfur-rich regions of the cuticle, specifi cally the A-layer and exocuticle. REFERENCES (1) C. Robbins, Infra red analysis of oxidised keratins, Textile Res. J., 37, 811 (1967). (2) R. E. Noble, Uptake of calcium and magnesium by human scalp hair from waters of different geo- graphical environments, Sci. Total Environ., 239, 1-3, 189–193 (1999). (3) J. Jachowicz, Hair damage and attempts to its repair, J. Soc. Cosmet. Chem., 38, 236–286 (1987). (4) J. M. Marsh, J. Flood, D. Domaschko, and N. Ramji, Hair coloring systems delivering color with re- duced fi ber damage, J. Soc. Cosmet. Chem., 58, 495–503 (2007). (5) G. R. Bhat, E. Lukenbach, and R. R. Kennedy, The green hair problem: A preliminary investigation, J. Soc. Cosmet. Sci., 30, 1–8 (1979). (6) K. V. Curry and S. Golding, Hair lipids. I. Extraction of fatty materials from hair clippings, J. Soc. Cos- met. Chem., 22, 681–691 (1971). (7) G. Chittleborough, A chemist’s view of the analysis of human hair for trace metals, Sci. Total Environ., 14(1), 53–75 (1980). (8) M. Villian, V. Cirimele, and P. Kintz, Hair analysis in toxicology, Clin. Chem. Lab. Med., 42(11), 1265–1272 (2004). (9) J. Bacso, L. Sarkadi, and E. Koltay, On endogenous and exogenous calcium content of hair samples used in XRF and PIXE measurements, Int. J. Appl. Rad. Isotopes., 33, 5–11 (1982). (10) I. M. Kempson and W. M. Skinner, Advanced analysis of metal distributions in human hair, Environ. Sci. Technol., 40, 3423–3428 (2006). (11) J. N. Audinot, S. Schneider, M. Yegles, P. Hallegot, R. Weneg, and H. N. Migeon, Imaging of arsenic traces in human hair by nano-SIMS 50, Appl. Surf. Sci., 231–232, 490–496 (2004). (12) I. M. Kempson, W. M. Skinner, and P. K. Kirkbride, Calcium distributions in human hair by ToF- SIMS, Biochim. Biophys. Acta, 1, 1624 (2003). (13) C. Merigoux, F. Briki, F. Sarrot-Reynauld, M. Salome, B. Fayard, J. Susini, and J. Doucet, Evidence for various calcium sites in human hair shaft revealed by sub-micrometer X-ray fl uorescence, Biochim. Bio- phys. Acta, 1619, 53–58 (2003). (14) A. Benninghoven, F. G. Rüdenauer, and H. W. Werner, Secondary Ion Mass Spectroscopy: Basic Concepts, Instrumental Aspects, and Trends (John Wiley & Sons, New York, 1987). (15) J. C. Vickerman, A. Brown, and N. M. Reed, Secondary Ion Mass Spectrometry: Principles and Applications (Clarendon Press, Oxford, 1989). (16) P. Hallegot and P. Corcuff, High spatial resolution maps of sulfur from human hair sections an EELS study, J. Microsc., 172, 2, 131–136 (1993). (17) C. Collin, B. Gautier, O. Gaillard, P. Hallegot, S. Chabane, P. Bastien, M. Peyron, M. Bouleau, S. Thi- baut, F. Pruche, A. Duranton, and B. A. Bernard, Protective effects of taurine on human hair follicle grown in vitro, Int. J. Cosmet. Sci., 28, 289–298 (2006).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)