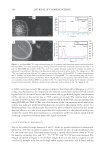
Cu AND Ca UPTAKE IN COLORED HAIR 345 (18) P. J. Heard, K. A. Feeney, G. C. Allen, and P. R. Shewry, Determination of the elemental composition of mature wheat grain using a modifi ed secondary ion mass spectrometer (SIMS), Plant J., 30, 237–245 (2002). (19) J. A. Swift and B. Bews, Chemistry of human hair cuticle. II. Isolation and amino acid analysis of the cell membrance complex and A-layer, J. Soc. Cosmet. Chem., 5, 355–366 (1974). (20) E. O. P. Thompson and I. J. T. O’Donnell, Studies on oxidized wool, Aust. J. Biol. Sci., 12, 490–499 (1959). (21) L. J. Wolfram, K. Hall, and I. Hui, The mechanism of hair bleaching, J. Soc. Cosmet. Chem., 21, 873–900 (1970). (22) R. G. Wilson, SIMS quantifi cation in Si, GaAs, and diamond—An update, Int. J. Mass Spectrom., 143, 43–49 (1995).
J. Cosmet. Sci., 60, 347–352 (May/June 2009) 347 Tyrosinase inhibitors isolated from the roots of Paeonia suffruticosa HSIOU-YU DING, HANG-CHING LIN, and TE-SHENG CHANG, Institute of Cosmetics Science, Chia Nan University of Pharmacy and Science, Tainan (H.-Y.D.), R&D Center, Sinphar Pharm. Co., Ltd, I-Lan (H.-C.L.), and Department of Biological Science and Technology, National University of Tainan, 33 sec. 2, Shu-Lin St., Tainan (T.-S.C.), Taiwan Accepted for publication November 6, 2008. Synopsis The inhibition of mushroom tyrosinase by Paeonia suffruticosa root-derived materials was evaluated. Six tyro- sinase inhibitors were isolated by ethanol extraction, n-hexane, ethyl acetate, n-BuOH, and water partition, silica gel column chromatography, Sephadex LH-20, Lobar PR-8, and high-performance liquid chromatog- raphy methods, and they were identifi ed as kaempferol (I), quercetin (II), mudanpioside B (III), benzoyloxy- paeonifl orin (IV), mudanpioside H (V), and pentagalloyl-β-D-glucose (VI) on the basis of spectroscopic evidence. The inhibitory activities of compounds I to VI against mushroom tyrosinase were determined with IC50 values of 0.120, 0.108, 0.368, 0.453, 0.324, and 0.063 mM, respectively. The kinetic study indicated that all purifi ed inhibitors acted competitively for the L-dopa binding site of the enzyme, with an exception of compound VI, which acted non-competitively. INTRODUCTION Tyrosinase is a copper-containing monooxygenase widely distributed in nature. The en- zyme catalyzes the fi rst two reactions of melanin synthesis, the hydroxylation of L-tyrosine to 3,4-dihydroxyphenylalanine, L-Dopa, and the oxidation of L-Dopa to dopaquinone. This o-quinone is a highly reactive compound and can polymerize spontaneously to form melanin (1). Although the pigment melanin in human skin is a major defense mechanism against the ultraviolet light of the sun, the production of abnormal pigmentation such as melasma, freckles, age-spots, liver spots, and other forms of melanin hyperpigmentation can be a serious aesthetic problem (2). Hence, inhibiting tyrosinase activity and prevent- ing abnormal pigmentation has been the subject of many studies (3–4). Paeonia suffruticosa Andrews (Paeoniaceae), commonly known as “Mudanpi,” is an im- portant crude drug used in Chinese traditional medicine as an analgesic a sedative an anti-infl ammatory agent a remedy for cardiovascular diseases, extravasated blood, and Address all correspondence to Te-Sheng Chang.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)