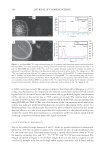
JOURNAL OF COSMETIC SCIENCE 354 determined by using the HPLC method. 8-Hydroxydaidzein was purifi ed as described in our earlier work (3) and dissolved in acetone as a stock with a concentration of 2 mg/ml. The stock of the compound was diluted 20-fold to a concentration of 100 μg/ml of either 50 mM pH 6.8 phosphate buffer or DMSO. Then, the diluted solutions, in 1.5-ml tubes covered with aluminum foil to avoid light, were placed on a bench at 25°C for 15 to 18 days. During the storage time, samples (20 μl) were taken out for HPLC analysis at the determined interval times. RESULTS AND DISCUSSION As shown in Figure 1A, when 8-hydroxydaidzein was dissolved in the pH 6.8 phosphate buffer, the compound was quickly degraded. In contrast, when the compound was dis- solved in DMSO, the compound remained stable at 80% after 18 days’ storage. Kinetic analyses showed that 8-hydroxydaidzein loss in both solvents was apparently the result of fi rst-order reactions (Figure 1B). The loss rate constants of 8-hydroxydaidzein in the phosphate buffer and DMSO were 4.48 × 10−3 and 2.5 × 10−4 hour−1, respectively. The above results show that the utilized solvent could dramatically affect the stability of 8-hydroxydaidzein in the solution. There are three possible reasons for the instability of Figure 1. Degradation of 8-hydroxydaidzein displayed by residual plot (A) or ln[Ct/Co] plot (B) in 50 mM of pH 6.8 phosphate buffer (♦) and DMSO (■). Samples (1 ml) were stored at 25°C, and 8-hydroxydaidzein was analyzed by HPLC. The HPLC analysis was performed on a Hitachi D-7000 HPLC (Hitachi, Ltd., Tokyo, Japan) system equipped with a L-7400 UV detector and a 250 × 4.6 mm i.d., ODS 2 Spherisorb C18 reversed-phase column (Phase Separation Ltd, Deeside Industrial Park, Clwyd, UK). The operating condi- tions were as follows: solvent, 30% acetonitrile/water containing 1% acetic acid fl ow rate, 0.8 ml/min detec- tion, 262 nm injected volume, 20 μl from 1-ml assay system containing 100 μg 8-hydroxydaidzein/ml of either DMSO or phosphate buffer. The residual 8-hydroxydaidzein of each sample at the determined interval times was calculated by fi tting the HPLC area of the sample to a standard curve of 8-hydroxydaidzein. Con- centrations ranged from 0 to 100 μg/ml.
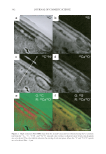
STABILITY OF 8-HYDROXYDAIDZEIN 355 Figure 2. Degradation of 8-hydroxydaidzein in a series of buffers with different pH values. The buffers used were 50 mM acetate buffer for pH 5 (♦) and pH 6 (■) 50 mM phosphate buffer for pH 7 (▲) and 50 mM Tris-HCl buffer for pH 8 (—) and pH9 (●). The experimental conditions were the same as those in Figure 1. the compound in the pH 6.8 phosphate buffer. One is that the compound might react with phosphate and result in instability. Another reason is that the compound was actu- ally not soluble in the tested water system and cannot be detected by the HPLC method. The last one is that the pH of the solvent could affect the stability of the compound. The fi rst two reasons were quickly ruled out because similar instability phenomena of the compound were also found when the phosphate solvent was replaced by 50 mM of pH 6.8 Tris-HCl buffer and the 8-hydroxydaidzein solutions prepared by either phosphate or Tris-HCl buffer were not found to produce any precipitation after centrifugation with 12,000 rpm for 10 min. Hence, the most likely factor that caused the instability of 8-hydroxydaidzein in the solution is the pH of the solvent used. To prove the point, ex- periments similar to those described above were repeated, but using buffers with different pH values to prepare the 8-hydroxydaidzein solutions. The tested buffers included 50 mM acetate buffer for pH 5 and pH 6, 50 mM phosphate buffer for pH 7, and 50 mM Tris-HCl buffer for pH 8 and pH 9. The stability of 8-hydroxydaidzein in different solutions was analyzed by the HPLC method. The results are shown in Figure 2. When the compound was dissolved in Tris-HCl buffers of pH 8 and pH 9, it was quickly degraded within 10 hours. In contrast, the compound remained stable above 85% after 20 days’ storage in acetate buffers of pH 5 and pH 6. In addition to the HPLC analysis of residual 8-hydroxydaidzein, the residual bioactivities of the 8-hydroxydaidzein solutions with different pH values were also determined at the end of the storage experiment. As shown in Figure 3, tyrosinase inhibitory activity and DPPH-radical scavenging activity of the solutions remained at 94% and 82% in the pH 5 acetate buffer and at 93% and 78% in the pH 6 acetate buffer, respectively, while no activity could be detected in the 8-hydroxydaidzein solutions prepared in the pH 8 and pH 9 Tris-HCl buffers. The results of both residual tyrosinase inhibitory activity and DPPH-radical scavenging activity of the prepared 8-hydroxydaidzein solutions with different pH values correlate well with those of residual 8-hydroxydaidzein in each solution. The results of the present study demonstrate that 8-hydroxydaidzein is unstable in alkaline solutions. CONCLUSIONS Thermal stability of soy isofl avones has been studied well in soy fl our (5), soy milk (6), and model solutions (7). However, little has been reported about the effects of pH on the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)