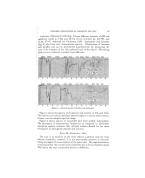
256 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 5ooo ooo ooo ,5oo, ,oo ,,oo ,,,o?,o, 90o [ i i i id-• •'1.1 i I'1 i i I I • '• • i i i i •'• i ['1 i i i'1 i 75O 70O i1'1111111 iiiiiiiii III IIIIiitl•:fl' IIIIIiiiiiJ-'1I ß I.i i i i i i i iJ- II I hi iii! iJ,.i II •1 I IJ/I I II I I-,P'r'l I I•Vl\l i i i i i l..d-I I i I I ,•i ! I I I I I I I I I I i j i i i i IIIllll Ill i I I I-III I I I I1/'11 I J i I I1•11 I I I I I I I I I I I I I I I I t I I I I I 1111 I I I I III • I I I/'11 I I I I I I III I I ! I I I I I I I I I I I I I/I I I I I II I II I ifil i iJ i i iii i i i i.,I i i i i I i i i/• • I I I I I I I i i ii/1•11 i i i i i i i i i i i iJ iiJ•l i i i i i i i'1 i j •]-•.. _ Ld • II I I I I I I I !ul IIII I I I I I I I I I I I I,IP, i/1111 I I I I I I I I,,,'r fllll I I I I II I I I I I Illfi I I I I I I I I I II Illlllll I I I i i i i i i i i i i i i i i i i.J/i IIIM IIJ"11111F I•11 I I i i i i iiiii i i i i i i i i i i I•'111 iii i i i i i i IIIii I/I iii111/1111 1• I I I I I I I I I IllIf I I I I I I I I I I I/1'1 1111 !,l I I I U L \,lllll lB I I I I I I I I I I I IIIII I I I I I I I I I I I111 I II I/',J I_11 I I I I I I I I I ,V't'q IIII.P'11111111 LI I I I I I I I I I IIIII I I I I I I I I I IIII I I II I I I/'1.1 i I I/ IIIIIIIIIIIil I I I I I I I I I I I IIIII I I I I I I I I I I ill I I I I I I./f I•1 I I I I I I I J/1 I I I I /"•/ C'r•/I I1•/•' I I I I I I• I I I I I I I I I I I I1111 I I I I I I I I I I III I I I I II ] I/ I I I I vlu [l[[[[],i[[[.[[][[[[[[[[.[•[ [• • •L,I CHLOROPHYLL II[ I I r I I I I I Illll I I I I I I I [ I Ill I I I IIll i,,.-k ! [[[ Lll I [ [ I I [ I [ [I]1[ [ I I [ I I [ [t]l It I I l] I I I I I I I\l/I I ] ' I I [ [ I I I [ ] I [ i I i ] i [ tl i I i ilp, i [ i [ [ i J Ii I[[[[Illll[11 111111111illrllllllll IIllllllllllllllilllfllllllll][llllt i 2 3' 4 5 6 7 8 9 l0 I I 12 13 14 15 5000 :30,00 20,00 1500 1300 O0 I0 f I I I I I I I'1 I I I I I I I I I I I I IJ-.d-q I I I I I I'1 I I I I I'1 I I I I I I I IIII111111 I III I III I IJ•rq/l•11 I I III I I I I III I I I III I I I I I I I I bl I I I I I I I I k•!•l I I I•1\1 I I I I I I I I I I I I I I I I I I I I I I I I I I IIII I I I I I I I.Z"-I I I I I L•l kl I I I I I I I I I I I I I I I I I I I I 1[11 II/111 I I II I ,i'-2"•1 I II I I/1• I•1 Ill II I I I I lll L_I-4 L-L• I I I I I I I V I/& I I I I I/Yl I I I I IA I I I I I I I I I I I I • "P'I I I I It"J / I I I I I I•,rll,d IM I I I III I t I I I../I I I I I F,l[111 I I I I L/I I I Iii H [ [• I [ I I I [ [If F/[ [ [ F'"'] [ i [ [ [ I•[ I [HJJ J I I•l [/l•r•!]l [ [[ I I Ill I [ I I I I Ill I I I J-/I I I i I I I Itl I [llll. ll I/[ I•/I I 1'4 I [ I I I I I t •11 I I I I I I II•.n I .v-I t ] I I I ] ] llkl 4Jlllfi III [/I rl I I [ I [ I I,,il I Illft.4' I I I l I [ I I I I ilrll }.11 iI•lJi IiL/I I I I I,l I I I I I I I I.r II IllIf I I I I I I I I I I I Iq 1/111M IlILq- • IN I L.KI I•l l']lJ• I I I I I I I I I I I I I• 11111\lfi,'l I I I I I I.l..,Ixl I/I I I I I IJ,'"l I I 1•4,11111 I I I I I I I I ] I I IWI II1',,li/1 Ik.l,.-rl I I I i i i i iiiii i i i i i i i i i i i i i i i I/ll ill i i i [ i i i i [ i i i I I I I 111 I1 II IIIJl, I I I I I I I I I I I I I I I,FI [i i i i i I i i i i i i i )0. 900 . 800 750 700 IIIIIIIIIIIIIIIIIIIIIIIIIIII I I I I I Iq [II I I I I I I I I I I IJ-I--• IIIIIl•1111zrlll] ß I I IJ./f I I I I I I I I I I I'-I,I I I•11 I I I I I1 •.-L,I.,'• I I I I. I I i I I I I I I IM.I./I I I I I I I H I I I I I I I I I I I I I I I I II'ill III I I• ill I Ill/ I I III IIIIIIII II1[• I I I I I I./11%LI J,-i-l-4d I I I I I I I I I I I I! I 1[ I,r-Yl• I I •i•,./I I I i"•l I I I I.l-• I rM'3rl • I•1•1/I I I I • • I'N[ I I•1-11 I•'-h• IJl/I/l I/l ITI• I I I I I I [ T/[ [I 1/I I'l I I I I I I I l] I'¾l I I I I I Ill I/I I MI/I I I I I I I I 1 I I I I I'l I I I I I [ [1 IIII111 IIIIIIIIII LIIIIIlIJJJ •JJ WATER SOLUBLE P•J-J-J-J-J-J-J-H Ill GHLOROPHYLLIN J J ,, J,, ,, J,' III ii iii1[ ii]•11111x[i Illlil]•] I :: 3 4 5 6 7 8 9 I0 I I 12 13 14 Figure 6.--Infrared spectra of water-soluble chlorophyllin and oil-soluble chlorophyll. I 6 ? 8 9 I0 II 12 [3 14 15 I 6 7 8 9 I0 II • • 14 15 Figure Z--Infrared spectra of olibanum oll and olibanum oil adulterated with 20% diethyl phthalate.
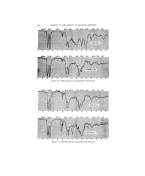
ß INFRARED APPLICATIONS OF COSMETICS AND OILS 257 ß ?:. Although the number of applications of infrared spectroscopy in this :ij::field are unlimited, such as easy detection of adulteration (Fig. 7 shows the i:':: :! adulteration of olibanum oil with 20% diethyl phthalate, which may be ?,.added to retard evaporation of odorants), identification, analysis of mix- '75': tures, control of raw materials and plant production, only three typical •i!i:i problems will be discussed in order to illustrate the analytical approach ,i .'i. made possible through the use of infrared spectroscopy. ii•: (a) Methyl-lonones ?:11: Figure 8 shows a series of alpha-ionones and the commonly used gamma- . •onones. All of these were of generally accepted degree of purity as 'obtained from various sources. The significant differences in intensities of bands at various wavelengths indicate that none of these materials is 100% 5 pure and contains more or less isomeric compounds and intermediate products from the processes of purification. ':"' (b) C-10 Essential Z%ohols :':' A very frequent problem in the essential oil industry is the evaluation :.: of the C-10 alcohols as shown below. :' Citronellol CHa--C-- CH=--CH•_--CH=--CH-- CH2--CH=--OH : CH• CHa OH Linalool CHa--C•-CH--CHr--CH•-- C--CH= CH2 ?: CH3 CHa Geraniol CHa-- C•--CH--CH=--C H2--C• CH-- CH=--OH (trans.) CIH= I CH, Nerol CHa--C•CH--CH=--CH2--C=CH--CH2--OH (cis) I CHa Rhodinol CHa--C=CH--CH•--CH•--CH--CH•--CH•--OH CHa CHa Figure 9 shows spectra of fairly high degree purity samples of these compounds. The location of infrared absorption bands from these materials can be assigned to the following functional groups and vibrations in these molecules. Figures 10 and 11 show how some commercial grade citronellols, rho- dinols, and geraniols vary in composition of isomers and impurities present. From the preceding band assignments, one can draw the following con- clusions: 1. Terminal unsaturation, such as in linalool, can be readily differen- tiated from double bonds in the chain of the molecule at several useful wave- lengths. 2. Tertiary and secondary hydroxyl groups will also give characteristic
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)