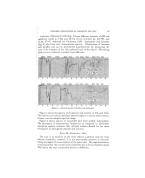
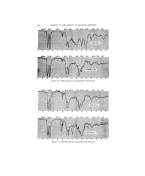
THE EVALUATION OF SOME ORGANIC SULFUR COMPOUNDS IN COLD WAVES*4- By ROBERT E. SAUTE, GLEN J. SPERANDIO, AND H. GEORGE DEKA¾ Purdue University, School of Pharmacy, Lafayette, Ind. THe. ART of curling hair dates back as far as ancient Egyptian times, when the "mud method" was used (1). The wooden sticks used in this method were soon replaced by metal rods (2), and as time progressed the rods were heated. This was the principle of the curling tongs which were still popular in the thirties. The first permanent waving method intro- duced into the United States originated in France and came here via P. Victor Senegas in 1884. This method included the use of concentrated sea water and heat. In the 19th century Marcel Grateau (1852-1936) de- veloped the famous method of waving the hair which bears his name. All previously used methods were soon abandoned when Charles Nessler, in 1906, devised a process for softening the hair by means of borax (3), and the introduction of mercaptans, first as depilatories, by Evans and Mc- Donough gave rise to their use as cold-waving agents. The substituted mercaptans have been found to be preferable in curling the hair because they are more efficient and have a less objectionable odor (4). Patents 2,577,710 and 2,577,711 covering the use of mercaptans in permanent wav- ing and cold waving were issued to McDonough this year. In 1922 the Croquinole method of winding hair was patented by Joseph Mayer, a Czechoslovak hairdresser, and introduced in the United States in 1926. Permanent waving of hair is caused by a change in molecular configura- tion of the keratin molecule, giving to the hair a lasting curl which cannot be changed by physical means. This change in configuration can be ac complished by the use of high temperatures, or by chemicals such as alkalis and certain sulfur compounds adjusted to a proper pH, in bases having a dis- sociation COLtant of less than 5 X 10 -a (5). Water curls and temporary sets differ frona permanent waves in that the curls are formed when unstable cross-linkages are present, and tend to re- turn to their normal configuration when moistened. The hair in its normal state is made up o}' regularly folded polypeptide chains with cystine, hydro- * This address was originally presented at Centennial Meeting of American Pharmaceutical Association, August, 1952, Philadelphia, Pa. 'f Presented at the December 11, 1952, meeting, New York City. 270
ORGANIC SULFUR COMPOUNDS IN COLD WAVES 271 ,. :. gen, and polar or salt .linkages, and in order to permanently curl the hair ':•" one must split the disulfide bonds (6). Before the chemical can split these disulfide bonds it must penetrate the cuticle of the hair. This sub- : jeer is covered in some detail by Reed, Den Beste and Humoller (7). Believing that a need exists for improved forms of cold wave preparations, ::i this investigation was conducted with the following objectives in mind: •::':. 1. To test the hair-curling power of some organic sulfur compounds at -: room temperature, in aqueous solutions adjusted to an optimum pH. 2. To formulate a cream base suitable for these chemicals and easy to •!':• apply to the hair. 3. To determine the relative toxicity of the chemicals in a cream base ..• on rabbits and human beings. 4. To compare the efficiency of these chemicals in a cream base and in ' specially prepared aqueous solutions. :• : 5. To compare the products formulated with cold waves already on the market with and without the use of a fixative. Six chemicals were obtained from Eastman Kodak Company: thiourea, ß cysteine hydrochloride, tautinc, mercaptosuccinic acid, •-mercaptopro-. : pionic acid, and thioglycolic acid.. The th-ioglycolic acid and .beta-met- i:: c.aptopropionic acid were purified by the use of a Todd Precise Fractiona- uon Assembly. Taurine and mercaptosuccinic acid were purified by re- crystallization. Aqueous solutions of these chemicals were made and ad- justed to a pH of 9.5 with ammonium hydroxide solution and allowed to stand for twenty-four hours. Cysteine hydrochloride showed a precipitate upon standing. These solutions were tested on human hair obtained from a local beauty shop and divided into locks of about 500 hairs each. One end of each lock was dipped in a melted mixture of equal parts of carnauba and ceresin waxes. The hair was washed with a commercial cream shampoo and solu- tions of the chemicals were applied to the hair with a pledger of cotton. The dampened hair was then wound on cutlers, again saturated with the test solution, and allowed to stand for forty-five minutes. At the end of this period the curls were saturated for five minutes with a 9% acetic acid solution as a fixative, then blotted with absorbent paper, again saturated with the fixative and allowed to stand for fifteen minutes, and again blotted. The hair was then carefully removed from the cutlers and immersed in the fixing solution before rinsing with tap water. All the solutions showed some curling power except the cysteine and thiourea, which were rejected for any subsequent t4sting. In formulating a base for the cold wave, the non-toxic, non-ionic Tween 20 and Span 20 were used. Amerchol was used in place of fanolin because
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)