JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS tergents, if it could be modified so that it would foam and could be removed from the hair when used with hard water. Thus, began another phase of the shampoo industry--a return to a soap base shampoo, modified with various synthetic detergents, new lime soap dispersants, and polyphos- phates and with variations to meet the requirements of dry, regular, and oily hair. The use of amine soaps in these shampoos has been increasing steadily, not only because they are easily prepared, good detergents, and mild, maintaining a low pH in the shampoos, but also because they are compatible with the various additives used to modify a soap shampoo. Amines are organic bases and the soaps of those which are used for sham- poos are formed by the reaction of amine and fatty acid in theoretical pro- portions--an addition reaction without evolution of water, but with a slight heating when the fatty acid and amine are stirred together. No external. heating is required for the reaction so that preparatior/s made with low titer fatty acids can be conducted quickly and easily at room temperature. Triethanolamine and monoethanolamine are preferred to other soap-form- ing amines for use in shampoos. Triethanolamine is a viscous, hygroscopic liquid with a slight ammoniacaI odor. Its alkalinity is less than that of sodium, potassium, or ammonium hydroxides and it is not harmful to tex- tiles n'or caustic to the skin. Monoethanolamine is a slightly viscous, hygroscopic liquid having a distinct ammoniacal odor and about the same strength as ammonia as a base. Triethanolamine oleate is a practically neutral soap of petrolatum con- sistency, completely soluble in water and having good detergent and emulsi- fying properties. Its water solutions are only mildly alkaline, with a pH of about 8, and moreover, they act to lower the alkalinity of sodium and potas- sium soap solution because the milder triethanolamine is the base finally liberated upon hydrolysis. Monoethanolamine oleate is similar to trieth- anolamine oleate in consistency and water solubility, but it has better de- tergent and lathering properties and a better toleration of hard water Water solutions of this oleate have a pH slightly lower than solutions containing' an equivalent amount of sodium or potassium oleate, but a slightly higher value than an equivalent concentration of triethanolamine. Amine soap shampoos are easily prepared by merely mixing the ingredi- ents together at room temperature. Since amines are weak bases, they will not saponify an oil or fat and must be used with a fatty acid to produce a soap. , Filtering the shampoos is rarely necessary, as a clear liquid should be obtained if no unsaponified or unsaponifiable material is present in the fatty acid and no free fatty acid remains in the shampoo. The presence of a little free amine prevents the latter property and improves the rinsing properties of the shampoo without increasing the pH appreciably. The choice of fatty acids is somewhat dependent upon the preference of
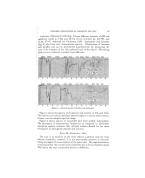
? AMINE SOAP HAIR SHAMPOOS 285 •?ihe manufacturer, but liquid fatty acids are used most generally because of ili:!11 the lower viscosity and quicker foaming properties of their soaps. A com- :i::bination of coconut oil fatty acid or technical lauric acid and oleic acid is ?suggested for amine soap shampoos--the former to produce a quick, copious •i?•! lather and good cleaning properties the latter to produce a finer foam and ?ii•to reduce any irritating effect the coconut oil soap may have upon the skin. :i :'i":: ' Very good shampoos can be made with triethanolamine soaps, but a iili•i noticeable improvement is attained by using a combination of mono- and 71 triethanolamine soaps. The mono soaps seem to improve the detergent !:i: and lathering properties and hard water toleration of the shampoos, and a :i(+.solution of the two soaps has the same mild action and low pH as that of a •i? triethanolamine soap. Results of rabbit eye tests with shampoos contain- i. :.i: ing equivalent amounts of soap revealed that a potassium soap shampoo is ':: more severe in action than a triethanolamine or a mono-triethanolamine 7:• soap shampoo, both of which are practically identical in degree of action. ::,. Potassium, triethanolamine, and monoethanolamine soaps were com- ::'pared by several methods to determine the effect of hard water upon their -foaming or lathering properties. The shampoo concentrates (Table 1) were formulated to contain the same amount of fatty acids, with sufficient amine or potassium hydroxide to neutralize the acids, and the propylene glycol content varied to produce the same total weight of shampoo concen- trate pep gram of fatty acids. TxBi. E 1 Parts by Weight for 400 Gin. Shampoo No. 1 No. 2 No. 3 No. 4 Oleic Acid 112.0 112 112.0 112.0 Coconut Oil Fatty Acids 84.0 84 84.0 84.0 Triethanolamine 116 58.0 ... Monoethanolamine 'i• 18 ... 24.9 Potassium Hydroxide •i :5 Propylene Glycol 1•:2 '•} 15i:1 152.5 The 400 gin. of concentrate was diluted with 1200 gm. of distilled water so that each test shampoo contained 122.5 gin. fatty acids or 195 gin. triethanolamine soap or its equiv•alent of other soap per liter. One com- parison of the effect of hard water upon shampoo lather was made by adding shampoo, a small portion at a time, from a burette to 100 mi. of hard water contained in a 300-ml. flask, agitating by shaking vigorously for 10 seconds, and continuing until the formed foam showed no signs of breaking in 10 seconds, but just started to break in 15 seconds. The agitation and foam stability times in such a test are selected arbitrarily, but should be main- tained for the series and preferably made by one operator. The amount of
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)