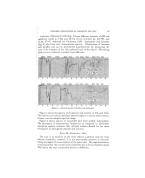
AMINE SOAP HAIR SHAMPOOS* By HELE• E. WASSSLL Carbide and Carbon Chem., Corp., Fellowship, Mellon Institute, Pittsburgh, Pa. H^•I• SH^•tvo0s achieved their original popularity because soft soaps were easier to apply, lathered faster, and were removed more easily from the hair than cake soaps. Clear liquid shampoos replaced soft soap when coconut oil became available in quantity at a reasonable cost. Better lathering and rinsing properties were attained with the coconut oil soap and the phosphates or carbonates, usually added to maintain clarity of the shampoo, assisted in the soap's toleration of hard water. Other oils were *Presented at the May 15, 1953, Meeting, New York City.
?' AMINE SOAP HAIR SHAMPOOS 283 iiiiused with coconut oil when it became apparent that a straight coconut oil ?11soap had a tendency to irritate some skins. These shampoos served their :i. purpose for a number of years, even though they had a fairly high pH and !i iWere not satisfactory when used with hard water. ?:i•: Ethanolamine soaps were suggested around 1930 as a replacement for }i Potassium soaps in shampoos. The shampoos could be made very easily ? 5and quickly, since no external heating is required to produce the amine i:•soap, and they enjoyed a slow but rather steady growth because of their ?i•.good detergency and mild alkalinity--their solutions having a pH of about i•.i18. While they tolerate hard water better than do potassium or sodium !).:soaps, they were not entirely satisfactory when used with very hard water. ?. Soapless shampoos of the sulfonated oil, non-lathering type were intro- :?duced about this time because of their bland nature and good rinsing prop- ii:-)erties. However, foam and detergency are usually synonymous in a ::..: woman s mind--even though one may not be contingent upon the other-- !'!: and this type of shampoo was never widely accepted. Synthetic detergents i!!:!:and wetting agents were being developed and soapless shampoos of the . 'i?lathering type soon began to appear on the market. Some of these synthe~ :i' uc agents seemed to be the answer for the perfect shampoo--quick, copious lather good detergent and emulsifying properties and soluble in hard ß •:water. The shampoos gained popular appeal very quickly and it began to • look as though synthetic detergent shampoos would replace most of the .: •i•i. soap shampoos on the market. Some of us who were evaluating solutions ::i:':11of these agents for various industrial applications soon realized that the solutions had a drying effect upon the skin and frequent use of a lanolin ii! lotion was necessary to keep the hands in good condition. This raised the !'•': question as to what effect continued use of shampoos, containing the syn~ ß 7:thetic detergents, would have upon the hair--in other words, are they such effective detergents and emulsifying agents that they will not only cleanse : the hair and scalp, but also remove too much of the natural oil? Our first answer to this question came in the form of requests from shampoo manu- facturers for a method to incorporate lanolin or oil in synthetic detergent shampoos without reducing lathering properties. They evidently hoped that the lanolin or oil would be selectively absorbed by the hair at the same time that the detergent was removing dirt and oily material from the hair. There were also reports that some beauty parlors preferred to use soap shampoos just prior to giving a permanent wave and that some operators would not give a permanent wave on hair that had been shampooed a num- ber of times with a synthetic detergent. It gradually became apparent that synthetic detergents did not produce the ideal shampoos. Although they could be used in all kinds of water, the hair became too dry and difficult to groom. Many manufacturers began to realize that soap would make a better hair shampoo than synthetic de-
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)