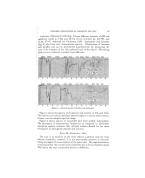
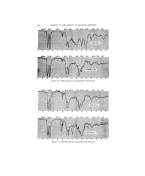
286 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS shampoo, required to produce the foam of described stability in 100 ml. of water of 180 to 720 p.p.m. hardness, seemed to be a logarithmic function rather than that 360 p.p.m. water required twice as much soap as 180 p.p.m. water, etc., to produce equivalent foam stability. Th.e results given in Table 2 indicate that a little more triethanolamine soap is required than monoethanolamine soap, but less than potassium soap, to maintain foams of equivalent stability in the hard water. TABLE 2 Shampoo (1 Conc. q- 3 HzO) Amount Shampoo in MI. to Produce Foam of Equivalent •Stability in 100 MI. Water Containing Hardness of:*--, 180 p.p.m. 360 p.p.m. 540 p.p.m. 720 p.p.m. No. 1 (MEA) 1.15 2.0 2.7 3.2 No. 2 (TEA) 1.30 2.2 3.2 3.9 No. 4 (K) 1.45 2.6 3.6 4.3 * Hard Water: Concentrate = 3.7 gin. MgCI2-6 H20 q- 6 gm. CaCI2 to 1 liter with dis- titled water. Water of 360 p.p.m. (CaCOa) hardness = 5 mi. concentrate q- 95 mi. distilled water. Two methods were used to compare the amounts of foam and/or lather produced by a given amount of shampoo in distilled and hard water. The Ross-Miles Pour Foam test was used because it is one of the few standard- ized comparative methods, but it seemed to provide a better comparison of dishwashing or laundry detergent foams than of shampoo foams or lathers. It is difficult to reproduce, by any laboratory method, the lather obtained from a shampoo applied to wet hair. However', the foam or lather, ob- tained by agitating 100 mi. of diluted shampoo in a quart bottle for I0 seconds, seemed to produce a better comparison for hair shampoos than the Ross-Miles method, where the foam is obtained by pouring the shampoo solution from a height of about three feet. No. 3--Ross-MILEs Povv. FOAM T•s'r Soaps Min. Foam on 5% Shampoo (0.975% TEA Soap or Its Equivalent) in: Distilled Water 250 P.p.m. H,_O 500 P.p.m. H=O Potassium 165 178 165 Monoethanolamine 171 178 165 M EA-TEA 165 152 140 Triethanolamlne 171 146 108 The results given in Table 3 indicate little difference in the amount of foam produced by any of the shampoos in distilled water, but as the water hardness was increased, the amount of foam produced by the trierhanoi- amine soap was decreased while the amount produced by the mor•oetha- nolamine or potassium soap was not noticeably affected. The results ob- tained by the second method (Table 4) indicated that potassium, monoeth-
:• : AMINE SOAP HAIR SHAMPOOS 287 . •:j?.anolamine , and mono- and triethanolamine soaps produce about the same ':i•!:::arnount of foam in distilled water, while a triethanolamine soap produces a :}•little less foam. The amount of foam produced by potassium soap in hard , ::::water is greater than that produced by the amine soaps, but it is a larger ?bubbled, faster breaking foam. Monoethanolamine soaps produce con- i!iisiderably more foam in hard water than triethanolamine soaps, indicating ?one of the advantages attained by using a combination ofmono- and trietha- ::•?:nolamine soaps in hair shampoos. •i: :i :' TABLE No. 4--AMou,'qT Fox•,* ML., o• 100 ML. SOLUTIO• AGITATED I• :::::: Qui•x BO•Lt vo• 10 S:coNus :,.:? ::?.::: Shampoo in 100 Distilled Water 250 p.p.m. Water 500 p.p.m. Water M1. Solution 5 MI. 5 MI. 2 MI. 5 MI. 4 MI, Potassium Soap 825 730 320 470 340 t Monoethanolamine Soap 900 525 245 375 320 MEA-TEA 900 325 150 280 190 Triethanolamine 640 325 95 265 130 :,:':" * Amount foam measured from top of foam to original solution level, ,:.:, J Foam starts to break immediately--others are fairly stable. ::i: The lower combining weight of monoethanolamine makes it more eco- nomical than triethanolamine for many purposes. Some shampoo manufac- turers contend that monoethanolamine is only economical in a hair sham- : poo when the cost of the fatty acids is lower than that of the amines. How- .:. ever, they are calculating the costs on the basis of a pound of soap without :i: considering that 1 lb. of monoethanolamine oleate should be at least as ef- fective as 1.2 lb. of triethanolamine oleate in a shampoo. : The amine soaps are made liquid for the shampoo by the addition of a ." solvent. Propylene glycol is suggested in preference to alcohol because it ß , is an excellent coupling agent as well as a diluent and hence improves the detergent properties of the soap. A fluid product can be maintained at all times by adding the propylene glycol to the fatty acid before adding the amine. The elevation of temperature from the heat of reaction when amines and fatty acids are mixed together can be used to advantage to ob- tain quick solution of the soap in the dilution water of the shampoo. The temperatures will vary with the size of the batch and the container, but those obtained in making 400 gm. of shampoo concentrate in a 600-ml. beaker indicate the elevation which can be anticipated. TABLE 5mTEMPEKATURE OF SHAMPOO CONCENTRATE UPON INITIAL STIRRING (400 GM. IN 600-ML. BEAKEl'.) TEA Shampoo Concentrate (22% propylene glycol) 106 MEA Shampoo Concentrate (38,6% propylene glycol) 136 TEA-MEA Shampoo Concentrate (30.3% propylene glycol) 120
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)