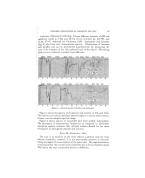
288 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS If the shampoo concentrate is diluted with at least an equal amount of water as sobn as it is made and while still warm from the heat of reaction, solution is attained quickly and easily and a better color is maintained than when the concentrate is stored for some time before dilution. The gradual discoloration of an amine soap shampoo has not seemed to prevent public acceptance as many formulators believed that it might. However, a great part of this discoloration can be prevented by the addition of 1 to 2 per cent of one of the polyphosphates. The phosphate also improves the shampoo's toleration of hard water. A good working basis for an amine soap shampoo concentrate is given in Table 6, which includes one mole each of oleic and coconut oil fatty acids and monoethanolamine, 1.05 moles of triethanolamine, and from 25 to 30 per cent propylene glycol. The concentrate can be adjusted with addi- tives and diluted with distilled water to suit the preferences of the formula- tor. Parts by Weight Coconut Oil Fatty Acids (C.W.--210) 210 Oleic Acid 282 Triethanolamine 149 Monoethanolamine 61 Propylene Glycol 282 If no inorganic soap (potassium or sodium soap) or salt (borax, tri- sodium, tetrasodium phosphate, etc.) is present, a simplified control test can be made to determine whether an ethanolamine soap or shampoo is neutral or contains free amine or free fatty acid. A distilled water solution of a weighed amount of soap or shampoo is titrated with standard acid, u:sing methyl orange as the indicator, to determine the total amount of amine present and a hot alcohol solution of another weighed sample is titrated with standard caustic, using thymolphthalein as the indicator, to determine the total amount of fatty acid present. All of the fatty acid in a freshly prepared amine soap will be titrated in the hot alcohol solution, be- cause mono- and triethanolamines are not indicated by thymolphthalein. If the required amount of standard acid is greater than the required amount of standard alkali, free amine is present and is calculated as trierhanoi- amine. If the amount of standard alkali is greater than the amount of standard acid, free fatty acid is present and is calculated as oleic acid. Synthetic detergents may be added to maintain adequate dispersion of lime so'aps and thus improve the rinsing properties of the shampoo, to im- prove the foaming properties of the soaps, and to increase the viscosity of the shampoo. The synthetic may be a nonionic and/or an anionic agent since the anionic soap will tolerate either type. The amount of additive
?/nay be varied to suit the requirements ofdifferent types of hair. However, iii'i.• the proportion of soap to synthetic detergent should always be high enough i) so that its foaming properties are increased rather than decreased by the i:'?'iaddition. There are also safety limits on the amounts of some additives ?. because of their irritating effect upon the eyes. i::ii:!' There are a number of synthetic detergents being used to improve the 71 iroperties of soap shampoos, but the majority of these fall into two general ß types of nonionic agents: (1) fatty acid alkanolamides or alkanolamine !.i fatty acid condensates, and (2) alkyl or alkylphenyl polyglycol ethers. )The fatty acid alkanolamides are somewhat less effective than the poly- •i!glycol ether type for maintaining dispersions of lime soaps, but more effec- tive for increasing the lathering properties and the viscosity of the soap ?Shampoo. Either or both types of these nonionic agents may be used with •:: the amine soaps in a shampoo. There are two methods of improving the rinsing properties of a soap ß .:shampoo: (1) by the addition of one of the described nonionic agents •'which will maintain an adequate dispersion of the insoluble lime soaps so :ßthat they can be removed from the hair by rinsing with water, or (2) by the addition of a compound which will prevent the formation of insoluble soaps. .i!11i The merit of these compounds, sequestering or chelating agents is indicated i by the rather rapid increase in the amounts used, even though they are a :51:i'i::comparatively expensive additive. The most efficient of these compounds :11i..appears to be the sodium salts of ethylene diamine tetra acetic acid. The :•i•i. pH of water solutions of the di-, tri-, and tetrasodium salts varies over a •:.: wide range and the salt selected for a given shampoo formulation will be i: "determined by the pH desired in the finished product. Some manufactur- ers may use both a nonionic and a sequestering agent in the same soap shampoo, adding the nonionic agent primarily for properties other than that of dispersing lime soaps. As stated previously, some of the fatty acid alkanolamides will increase ß the viscosity of an amine soap shampoo but, in some instances, the viscosity of the shampoo was not increased until a polyphosphate or a chelating agent was added. Thus, a shampoo, containing 20 per cent amine soap con- centrate and 8 per cent of one of the alkylolamides, had about the same viscosity as one containing only the soap concentrate. However, the ad- dition of 2 to 3 per cent of either sodium tripolyphosphate or one of the chelating agents produced a viscosity equal to a shampoo containing 50 per cent amine soap concentrate and 50 per cent water. It can be seen that, although the formulating of a good soap shampoo is not quite as simple as it was some years ago, the extra time and effort spent in developing a formula should be apparent in increasing sales of the product.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)