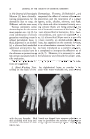
30 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS per cent of some dissolved substance. The answer is that the method is not sensitive in that sense--minimum amounts of 5% for recognition being typical also it is a tool for studying the solid portion of a system, not the liquid. POLYMORPHISM OF SIMPLE SATURATED TRIGLYCERIDES The polymorphism of glycerides has been recently and variously re- viewed (5, 6, 7). The views of Bailey and Lutton are in essential agree- ment but differ from those of Malkin. Triglyceride is, of course, the principal type of compound in fats. The single fatty acid triglycerides of which tristearin is typical have three crystalline forms or phases--alpha, beta prime, and beta in increasing order of stability, density, and melting point (see Table 1). T.•BLE 1--Cm•v,•CTER•STmS Or TmsT•.• POLYMO•V•S (6) Short Spacing Form Basis for Nomenclature fk. M.P.,ø C. L.S., •. Alpha 4.15 line 54 50.6 Beta prime 4.2 and 3.8 lines 64 46.8 Beta 4.6 line 73.1 45.15 The characteristic appearances of the diffraction patterns of tristearin are shown in the drawings of Fig. $. The so-called short spacings which give rise to the outer group of rings are related to the planar distances of separation of the rows of long parallel hydrocarbon chains which are a major portion of glyceride molecules the long spacings which give rise to the inner rings are related to lengths (or integral multiples of the lengths) of the long molecules. The alpha crystalline form is obtained by chilling the melt, beta by crystallization from solvent or transformation of the metastable forms at elevated temperatures. The somewhat elusive beta prime can best be ob- Alpha Beta Prime Beta Figure 5.--Main features of tristearin diffraction patterns.
THE POLYMORPHISM OF GLYCERIDES 31 rained by careful but not too slow crystallization from melt just above the alpha m.p. A notable feature, which seems to be quite common in a wide range of compounds, is that the alpha m.p. represents the approximate super- cooling limit. Under the polarizing microscope, alpha appears as a mosaic of small spherulites beta prime is commonly obtained as bright spherulites or from solvent as fine needles, while beta gives blades or plates (Fig. 4). Al- though alpha is invariably obtained on any rapid cooling of melt, it is fleeting. In compositions which are sufficiently different from tristearin, beta prime may be reasonably stable. Characteristically, in accord with their crystal habits, beta prime is more highly stiffening than beta. An important point, true of tristearin and widely applicable to polymor- phic substances, is that solid phase transformations occur much more rapidly in the presence of solvent (including oil). Tripalmitin is very similar to tristearin in polymorphism but with lower corresponding melting points. MIXED C•0-C•s SATURATED TRIGLYCERIDES Important deviations in solidification behavior from that of tristearin have been observed with the mixed palmitic-stearic triglycerides as seen in Table 2. TABLE 2--POLYMORPHISM OF MIXED C16-Cls SATURATED TRIGLYCERIDES (6) Triglyceride C.M.P., o C. Forms Other Than Alpha SPS 68.5 Beta only PSS 65.2 Beta prime from melt, beta from solvent PSP 68.6 Beta prime only SPP 62.7 Beta prime and beta The striking points are, of course, that SPS entirely lacks the highly stiffening beta prime form while PSP lacks beta. These two glycerides are something more than laboratory curiosities--SPS being a major component of completely hydrogenated lard and PSP being obtainable by way of the disaturated component of stillingia tallow (8). SIMPLE SATURATED DIGLYCERIDES The simple saturated diglycerides with one free hydroxyl group per glyc- erol group are of two types as exemplified by 1,3-distearin and 1,2-distearin. The former is much more readily prepared in a pure state and greatly pre- dominates in the equilibrium mixture (an equilibrium being presumed on the basis of monoglyceride behavior). Their polymorphic behavior is strikingly different as shown in Table 3.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)