42 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS tained. In addition, a fatty acid with a very high stearic acid content can be obtained by hydrogenation of oils such as soybean oil which contains a high percentage of C•8 unsaturated fatty acids, i.e., oleic, linoleic. and linolenic. As mentioned earlier, the major commercial stearic acid is one with a ratio of approximately 55 per cent palmitic and 45 per cent stearic acid. Depending upon the method of manufacture and quality desired, it will contain 90-98 per cent of the mixture of palmitic-stearic acids and up to 10 per cent oleic and myristic acids. The grades most frequently used for cosmetics are double or triple pressed stearic acid. Single pressed stearic acid with its relatively higher proportion of unsaturated fatty acids is un- suited for most cosmetic formulations. Among the foremost users of commercial stearic acid are the cosmetic manufacturers who have found that it best fits the requirements of the cosmetic products possibly because of its unique crystallization behavior. It provides the finished physical properties and performance character- istics which are difficult, if not impractical, to duplicate by other combina- tions of fatty acids. In the cosmetic field, uniformity of finished product characteristics is extremely important. Deviation in crystalline structure can mean varia- tions in appearance, body, texture, etc., of cosmetic creams and pastes. What are the reasons? There are so may factors involved it would be impossible to cover every detail that might be of interest within the limited scope of this review. With this in mind, a number of factors that are of basic interest have been selected. Perhaps a study of some of the physical chemical properties will help us understand them better and thus assist in pointing the way to obtain the desired properties in formulat- ing cosmetic preparations. CRYSTAL STRUCTURE AND MELTING POINT Commercial stearic acid has a well-defined crystalline structure. There is some evidence that this is due to the formation of an intermolecular compound of 1:1 palmitic-stearic acid in the composition range of typical commercial stearic acid. This crystalline structure is related to certain performance characteristics, such as melting point, shrinkage, hardness, toughness, and texture. Palmitic-stearic proportions outside this range will give different, perhaps inferior, crystalline structure, melting point, texture, etc., in the cosmetic preparations. To illustrate this phenomenon, let us examine the typical liquidus (melt- ing point) curve for the binary system palmitic-stearic acid (1). See Figure 1, "Mol per cent Stearic Acid vs. Melting Point." From this curve, it is seen that as the proportion ofstearic acid in palmitic is increased up to about 30 mol per cent the melting point is lowered from
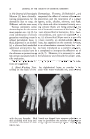
PHYSICAL CHEMICAL PROPERTIES OF STEARIC ACID 43 •0 ß 68 o 66 • 64 • 62 • 6o • 5s [• 56 54 0 10 20 30 40 50 60 70 80 90 100 MOL % STEARIC ACiD Figure 1.--Mol % stearic acid vs. melting point. 0 10 20 30 40 50 60 70 80 90 100 WEIGHT % STEARIC ACID Figure 2.--Weight % steric acid vs. crystal .qlZC. 62.9øC. to about 55øC. This is the point of lowest melting for any mixture of these two fatty acids and is ordinarily referred to as the eutectic. As the stearic acid proportion is increased to about 50 mol per cent, the melting point gradually rises to about 58øC. Commercial stearic acid containing 55 per cent palmitic-45 per cent stearic (weight basis) falls in this section of the curve. On a mol basis this is equivalent to about 57.5 per cent palmitic-42.5 per cent stearic as there is only about 10 per cent difference in molecular weight between these two fatty acids. As the proportion of stearic acid is increased from 50 to 100 mol per cent, the melting point increases sharply to 69.9øC. From this curve it is apparent that mixtures in the range of approximately 70 per cent palmitic-30 per cent stearic acid to 50 per cent palmitic-50 per cent stearic (tool per cent) have properties different from those of mixtures containing higher or lower percentages. Other physical-chemical measurements are of interest and serve to sup- port the evidence obtained from the melting-point curves. SIZE OF CRYSTALS Pure palmitic and stearic acids have comparatively large-sized crystals. Under a given set of conditions, crystals of palmitic acid are larger than those of stearic acid. The relationship between crystal size of mixtures containing various proportions of palmitic and stearic acid is shown in Figure 2, "Weight Per Cent Stearic Acid rs. Crystal Size." Crystal sizes are given qualitatively as they were not determined by actual measure- ment. As the proportion of stearic acid in palmitic is increased from 0 to about 30 per cent (weight) the crystal size becomes smaller. At the eutectic they are so small they appear amorphous or noncrystalline to the unaided eye.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)