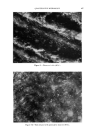
QUANTITATIVE MICROSCOPY 505 cation. In dilute solutions (10 -6 M) the cation exists as a monomer and fluoresces bright green. With increasing concentration dimerization sets in. The dimer has an emission spectrum with its maximum in the red spectral region, so that the fluorescence color indicates the dye concentra- tion. This so-called concentration effect has successfully (29) been em- ployed to distinguish between living and dead protoplasm. This applica- tion permits rapid testing of the effectiveness of antibacterial agents in skin preparations, deodorants or toothpastes by bacterial counts and replaces, when applicable, the time-consuming growing of colonies. To obtain re- producible results it is necessary to control the pH carefully with buffers, to use excess dye solution, and to stay within a certain pH-range. Also, it is necessary to check whether the tested preparations contain fluorescence quenching agents and whether the bacteria under examination show the typical green-red transition (Fig. 15). The uncharged molecules of several basic dyes accumulate readily in lipid phases. Addition of a fluorochrome to fatty substances allows determina- tion of the uniformity of very thin films of these substances, or their pres- ence, or their penetration. One of the great advantages of fluorescence techniques is, as has been mentioned before, the very high sensitivity of detection. One can clearly recognize a particle as stained when the concentration of dye in it is as low as 1:10,000. For particles smaller than 1•, which show up very clearly Figure 15.--Mucosa cells with bacteria stained with Acridine Orange (320X).
506 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS in a fluorescence microscope, this means that as little as 10 -•s g. of fluores- cing material can be detected. SUMMARY Problems of cosmetic research and development are highly complex and extremely diverse in nature. It is not the intent of this article to suggest certain microscopic techniques for the solution of specific problems nor to discuss in detail the intricacies and accuracies of any one of the described techniques. On the contrary, the article tries to show, with the aid of a few selected specific problems, a general concept of a systematic approach which could usefully be applied to a multitude of different problems. In this approach, the first step will be only qualitative, i.e., an attempt to make the process accessible to direct microscopic observation. This will very often be possible, if not directly, then in the form of models or simulated sur- roundings. The result of this observation will normally be that the method is found unsatisfactory. Modification in general preparative techniques and selection of a more suitable optical method are the next steps. This is followed by specific adaptations of the preparative technique to the selected and appropriate optical method. Nothing is more revealing than a direct visual observation of "what happens," and one picture is worth not only a thousand words but, at least in the beginning, quite a few automatic sensing devices. This is particu- larly true in the study of dynamics, for example, by means of movies taken through the microscope. The first attempt at quantitative analysis can now be made, encompass- ing a survey of all available parameters and investigation of each one of them. No hesitation should be shown to create concepts or parameters which describe directly observable facts or phases of the process under study, even if they have to be expressed in terms which are not normally found in the scientific literature. It could well be that the process cannot or has not yet been described quantitatively in physico-chemical terms such as cm./sec. or mol./sec. Numerical values for these terms can normally be obtained only after the process has been understood. From these developed parameters, one indicating parameter should be selected which shows the highest coefficient of correlation to the examined effect, and this correlation should be confirmed. Actually, the indicating parameter may not always be the most obvious one. In all of these proceedings, it is advisable to use relative values only so that each experiment carries its own reference standards. This approach is much safer in a field where side effects may not have to be discovered and where the influence of systematic errors is not established. In many cases, relative values are all that may be of interest for the moment, and these may
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)