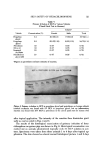
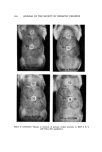
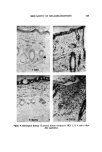
122 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS o 1 ---' 1% HCP in ACETONE --- 3 '"-- 5 """ 10 tt 2 3 4 5 6 7 8 9 10 11 12 13 14 DAY Figure 4. Development of primary irritant reaction (guinea pig) after topical application. Three days after the application, extravascular out- flow of erythrocytes was observed in the upper and middle layer of the der- mis. In addition, degeneration of the walls of minute blood vessels was also noticed. On the other hand, the epidermis was almost normal. Cellular infiltra- tion was not distinct in the dermis. Four days after the application, a marked cellular infiltration composed of small round cells, neutrophils, histiocytes, and a few eosinophils was observed in the upper and middle dermis. In addi- tion, blood vessels were dilated and an extravasation of erythrocytes was no- ticed. Collagen fibers were considerably edematous. In the lower layer of the dermis, blood vessels were dilated and affected with thrombosis. Degenera- tion of vascular walls and extravascular outflow of erythrocytes were also noticed. Figure 7 shows the results of examination of the animals for phototoxicity to HCP. In guinea pigs, the positive skin reaction was compared between the sites of HCP topical application alone and those of HCP topical application accompanied by ultraviolet irradiation. The results showed that positive skin reaction was always higher at the latter site, with HCP concentrations of 0.05, 0.1, 0.25, or 0.5% but not at 1%. In rabbits, the positive response was also greater at the irradiated site than at the nonirradiated site applied with 0.25% of HCP but the difference in response was less marked at 0.5 and 1.0% con- centrations of HCP.
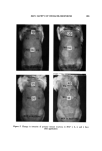
SKIN SAFETY OF HEXACHLOROPHENE 123 Figure 5. Change in intensity of primary irritant reactions to HCP 1, 2, 3, and 4 days after application
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)