174 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS between probable exposure of the skin or mucous membranes during normal use and the level of exposure which would produce an adverse reaction. In order to err on the side of safety, broad margins of safety are sought by exaggerating the levels and conditions of exposure in the test procedures. However, injury may result from grossly excessive direct con- tact of many tissues with even the most innocuous environmental chemicals and so a rational approach to exaggeration is essential. The degree of chemical insult that the skin or mucous membranes might be expected to tolerate is somewhat problematical. The extensive testing carried out on the safety of ingested materials, such as food additives, offers little guidance as such investigations are principally aimed at demonstrating toxic effects after systemic absorption. The aspect of safety evaluation for ingested substances corresponding to topical administration is the direct effect, if any, of the test materials on the gastro-intestinal lining. Severe irritation of the lining would indeed be observed under conditions of gross exposure to many universally-accepted food materials and especially con- diments such as vinegar and mustard. In other words, although there should be a wide margin of tolerance once a test material has been diluted in the body fluids following absorption, a narrower margin is to be expected in the case of a tissue in direct contact with the test material. Whilst acknowledging that there are likely to be considerable differences between direct exposure and exposure after absorption, it might be instruc- tive to consider the postulates on which the safety evaluation of food additives is based. A hundredfold safety factor (1) is commonly quoted this may be deemed to offer a tenfold allowance for the greater suscepti- bility to systemic toxicants of man compared to laboratory animals, to- gether with a further tenfold allowance for variation in susceptibility between individuals. In terms of systemic toxicity, such a tenfold allowance for inter-species differences in metabolic transformation and excretion seems reasonable. Effects on the skin and mucous membranes, however, are not primarily dependent on species-specific metabolic pathways. Indeed, the skin of those martahals most often used for irritancy testing approximates quite closely to human skin in its susceptibility to irritation or even shows greater sensitivity to some irritants (2, 3). The epidermal horny layer of the skin is an important barrier to the absorption of foreign chemicals (4) and provides the first line of defence against irritants. Thickness of the horny layer varies across the human skin and in some regions it is thinner than the horny layer of other mammals. However, human epidermis overall is much thicker (5) than in most
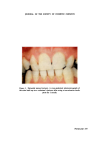
EXAGGERATED EXPOSURE IN PREDICTIVE TESTING 175 mammals, including the rabbit, rat and mouse this probably explains why applied substances do not easily penetrate human skin (6) and why, on the whole, it is no more susceptible to irritants than is the skin of these species. When irritancy tests are carried out on animal skin, it would therefore be irrational to allow a tenfold margin for interspecies differences. The possible irritancy of cosmetic materials in contact with tissues of the eye is usually studied by instillation into the conjunctival sac of the rabbit eye. On the basis of wide experience of such tests, Davies (7) suggested that the rabbit eye was decidedly more sensitive to irritants than the human eye. Thus no allowance for interspecies differences seems necessary for extrapolating rabbit eye test results in terms of hazard to man. A major factor involved in selecting the appropriate levels of exaggera- tion in irritancy testing is that a quantitative enhancement of responses may help to establish a meaningful safety margin, whereas a qualitative change in the type of response could well render the findings incapable of inter- pretation qualitatively atypical responses might well result from gross exaggeration of exposure levels in tests for skin and eye irritancy (Fig. 1). To ensure that testing procedures give the information required for safety assessment, a critical re-appraisal of current methods is needed. TEST METHODS In the study of systemic toxicity, suitably exaggerated dose-levels are administered to laboratory animals in the diet, by gavage or by injection. Exposure of the skin or mucous membrane to substantially exaggerated quantities of test material is seldom practical in the study of irritancy. Direct contact within a circumscribed area is essential an exaggerated quantity applied to a larger area will not necessarily intensify the response. Exposure to a raw material may often be exaggerated by applying con- centrated solutions, but this would not be feasible for complete formula- tions. Even with raw materials, unrealistic effects may occur, for example, owing to hypertonicity or grossly abnormal hydrogen ion concentration such exaggeration could well produce effects totally irrelevant to the hazards of ordinary use, by producing qualitative rather than quantitative differences in response. An alternative way of exaggerating topical exposure is to lengthen the time of contact compared with normal use or to make multiple applications. This is helpful if it influences the response quantitatively without provoking
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)