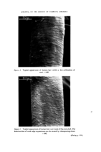
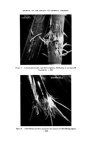
166 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Sulphuric acid conc. Sulphuric acid 4 M Hydrochloric acid 6 M NaOH 8 M Nitric acid 50•o Diphenylcarbazide 1 •o in ethanol Hydrogen peroxide 3•o Nessler reagent (K2HgI4) for ammonia Ammonium molybdate 0.5•o in conc. sulphuric acid Methanol Petroleum ether (40/60) Benzene Carbon tetrachloride Dimethylformamide Glycerol Borax Potassium bisulphate Potassium ferrocyanide Methods Additives should first be removed from the sample and the pigments isolated. There are three procedures to do this, which depend on the physicochemical properties of the sample. In these operations centrifuging is an important step, since the striated sediment will in general give visual information of the separated components. Removal of additives and isolation of the pigments. Procedure of removal of additives depends on the physicochemical properties of the sample: Hydrophilic (miscible with water): procedure A Lipophilic (immiscible with water): procedure B 'Wax cake' for instance mascaras: procedure C Procedure A. Mix 1 g sample with 40 ml water. Homogenize with blender or Ultra Turrax homogenizer. Centrifuge in 45 ml tube for 15 min at 3-4000 rev/min. N.B. The striated sediment will give valuable information, in particular, of which components the pigment-mixture consists. The analysis will proceed according to these assessed colour components of the dried sediment. The supernatant liquid might be coloured. If the colour can be adsorbed with Polyamide powder (procedure under Table III), the colour is in its water soluble form. If not the colour might be a suspension of small particles of dye-lakes, which is mainly present in the sediment. Procedure B. Mix 1 g sample with 20 ml petroleum ether. Heat gently on a water bath to defat the powder thoroughly. Decant the petroleum ether. Repeat the defatting procedure twice more. Dry the powder and suspend in 40 ml water. Proceed as under A. Procedure C. 'Wax cake' mascaras might contain chromic and iron oxides, and the different kinds of carbon blacks. For the identification of the carbon type
RISK-BEARING SUBSTANCES IN COSMETICS 167 method A or B must be used. For the identification of chromic or iron oxide the sample should be ashed on a platinum dish to remove additives and the carbon. The next step is to assess the colour components visually by observation of the original sample and of the striated centrifugal sediment. Assessment should be within the following defined colour groups: GREEN RED BLACK BLUE YELLOW-ORANGE PEARL VIOLET BROWN WHITE For example bluegreen should be assessed Blue and Green. The assessment of colour components of the sample is necessary to guide the analysis. Table H gives for each colour component the possible groups of colour compounds. Table II. Possible groups of colour-compounds for the assessed colour components GREEN Chromium-oxides Ultramarines Organic colours BLUE Ultramarine Iron-compounds Cobalt-compounds Organic colours VIOLET Ultramarines Manganese-compo unds Organic colours RED Iron-compounds Organic colours YELLOW-ORANGE Iron-compounds Organic colours BROWN Iron-compounds Manganese-compounds BLACK Carbon Iron-compounds PEARL Pearlescent compounds WHITE White compounds All the possible groups of compounds should now be investigated systematic- ally by characteristic reactions and methods that belong to each group of com- pounds. Characterization reactions and methods Ash. Ash a small sample on a platinum dish, heated by a microburner. Observe the colour changes during the heating and after cooling. Sulphuric acid 4M. Add a small amount of the isolated pigment to -•- ml of the acid. Observe before and after gentle heating. Gas evolution and colour changes
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)