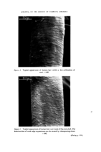
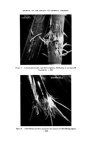
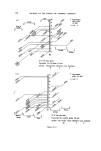
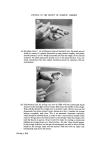
RISK-BEARING SUBSTANCES IN COSMETICS 197 tic Sam •Ie no. o oo o oo • o o ß 00¸ 0 S tort el oO ß ß g lc: relahve retenhon hmes (Benzo(•mne:) 1,5 1 UI, 9 14,7 0,7 2 • 2 ,G I 5,30 "51 D 1'9 14,7 0,72 • 2,6 0,600 i 00,90 D 5,21:] 10,72 i]1,3 rl 2,6 i i i i 511• Start 1,0 2,0 5,0 4,0 5,0 IO, 72 1,5 II, I 4,8 I I,,5 12,7 I Figure 10. Example of tlc/glc matching for the identification of sunscreens. Sample no. 10 contains B (Givtan F). Sample no. 11 contains A (Eusolex 3573) and possibly C (Solprotex 1). Sample no. 12 contains B (Givtan F). Sample no. 13 and 14 contain A (Eusolex 3573) and possibly C (Solprotex 1). Diisopropylether-n. Hexane-Acetic acid 75 ß 35 ß 1, by volume. Visualization was with Ehrlich reagent. Sample no. Rf 1 48 Benzocaine 0.55 + + p.Aminobenzoic acid 0.45 -- + Glycerol ester of pAB 0 -- + Saponification of both samples, followed by extraction and tic, left only one spot of the free acid. Procedure: Boil « g of the sample with 5 m183/0 ethanolic KOH during 30 min. Cool and dilute with 20 ml water. Acidify with 4M HC1 to a pH of 1-3. Extract the free acid with 2 x 10 ml chloroform. Evaporate the chloroform fraction to 1 ml, and use this for tlc. Benzocaine was isolated from both samples by means of column chromato- graphy. The isolated fractions of Benzocaine (Fraction I, 4 of sample no. 1 and Fraction I, 3 of sample no. 48) were confirmed by its ir spectra and its response on glc. Procedure: Use 2 x 20 cm glass columns. Fill with 15 g Silicagel Merck (diam. 0.05-0.20 mm) suspended in n. Hexane. Add a mixture of (1 g sample + 1 g Na.,.SO• exsicc. + 5 ml n. Hexane) on the column. Elution proceed as described below. The column fractions are concentrated and controlled by tlc (as above).
198 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Dihydroxyacetone in suntan preparations The browning agent dihydroxyacetone can be detected on tic plates of cellulose by the same method as for the separation of sugars. It is interesting to note that with one of the samples this tlc system shows the presence of a mixture of synthetic organic colours, namely a grey, a red and a yellow spot. Method. Extraction: Dissolve 1 g of the sample in a mixture of 0.5 ml water and 3.5 ml methanol. Mix thoroughly. Use aqueous methanolic layer for tlc. Reference solution: Dissolve 400 mg dihydroxyacetone in a mixture of 2 ml water and 23 ml methanol. (1 [tl = 20 •tg dihydroxyacetone.) Tic plates: Glass plates with 0.3 mm coating of cellulose MN 300. For the coating of 5 plates 20 x 20 cm: mix 15 g cellulose MN 300 with 90 ml water. Dry at 90øC until thoroughly dry. Solvent system: Used by Raadsveld and Klomp (27) for tic of sugars: Water- Ethylacetate-Pyridin (15: 60: 25, vol). Unsaturated tank. Time about 45 min for 15 cm path. Spotting: Extract 10 •tl reference solution 5 gl. Visualization: Spray with a mixture of: 1.3 ml HaPO4 85•o + 0.93 ml aniline + 100 ml 70•o ethanol. Then heat the plates at 100øC for 15 min. Dihydroxyacetone Rf 0.85: Tomato red Glucose: 0.30: Brown Rhamnose 0.60: Green. Results The analytical results on a range of commercial products are finally tabulated in Table VIII. In 20• of the samples more than one sunscreen is used in a product, and which might be present in both phases of the emulsion. From the tic data the conclusion can be drawn that many of the industrial sunscreens as offered by the chemical industry are not always single compounds, but mixtures of 2, 3 or some- times even 5 components. The browning agent dihydroxyacetone was found in 8 samples of 4 brands. Benzocain was found in 6 samples of 2 brands, but lidocain has not been detected. Due to an incomplete library of reference compounds several sunscreens remain unidentified. THE ANALYSIS OF PROPELLANTS AND SOLVENTS IN HAIRSPRAY AEROSOLS Aerosol cosmetics which take an important place in our society contain major proportions of propellants and solvents. These compounds have toxicity hazards if inhaled and could also be fire hazards because of the flammability of several of
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)