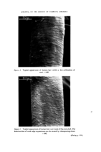
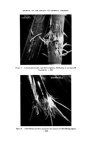
170 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS coloured, due to the formed manganate. Destroy excess peroxide by heating. Cool. Dissolve melt in 2 ml water. The colour is still green. Acidify. The colour turns into violet, caused by permanganate. Metal analysis. if burnt umber is confirmed, metal analysis should be made. The ratio Fe/Mn might give an indication of the umber origin. Co-compounds 77346 Cobalt aluminate. CoO.A1203 Ash. Cobalt aluminate is the only eye make-up colour that retains its beautiful blue colour during and after the ashing. Borax bead. The colour of the borax bead is deep blue. Carbon 77266 Carbon black 77267 Bone black --Vegetable black Organic colours 75470 Carmine. A1/Ca lake of carminic acid 75810 Cu-chloro- phyllene There are many synthetic organic colours possible. Pearl-pigments 77000 Aluminum powder 77400 Copper/ bronze powder Ash. The residue after ashing is of importance. Carbon black has no residue Bone black leaves 80•o of white residue. Vegetable black leaves a much smaller amount of residue. $ulphuric acid 4M. This reagent is only of importance to confirm the ash residue of bone black, which contain phosphates. A small amount of the ash residue is heated with « ml sulphuric acid 4M and NH4 molybdate reagent. The phosphates give a yellow coloration. $ulphuric acid 4M. Colouring of the dilute acid after heating might show the presence of an organic colour. Dimethylformamide. Colouring of this solvent by a small amount of the isolated pigment might show the presence of an organic colour. Filter. Concentrate by evaporation (in the hood !) on a waterbath and use the solution for tlc. N.B. Many lakes are appreciably soluble in dimethylformamide. Methanol. This solvent will dissolve the unsulphonated organic colours, leaving the lakes undissolved. Concen- trate the solution and proceed with tlc for identification. Thin-layer chromatography. Use the extract of 6 and 7 for tlc using the systems of chapter 3: identification of lipstick colours. Ash. Aluminum powder burns with flashes, leaving a white oxide. If there is a strong orange colour, BiOC1 might be present. This compound turns white again after heating for a short time, but if heating is excessive the colour remains yellow after ashing. TiOa and ZnO turns
RISK-BEARING SUBSTANCES IN COSMETICS 171 77820 Silver powder 77480 Gold powder BiOC1, as such or precipitated on mica TiO2 precipitated on mica or talc (Titanated mica) White-Blue-Red Green-Gold Guanin, pearl essence 2.Amino, 6.Oxy purine also yellow during the ashing, but not so intense as with BiOC1. The yellow colour turns white again after cooling. Sulphuric acid 4M will dissolve BiOC1. Then add Na•S. A brown precipitation of Bi2Sa occurs. Copper and bronze powder will dissolve with a blue colour, which deepens after addition of ammonia. HCl 6M. Titanated gold micas contain a little iron oxide. Heat it with a little HC1 6M, dilute with equal volume of water and add several crystals of K. ferro- cyanide. After some time a green-blue coloration occurs. NaOH 8M. This is an excellent reaction for Aluminum powder, which generates hydrogen. This reaction of A1 with a base is much better than with an acid. Guanin dissolves in NaOH 8M. Titanated mica retains its colour in this reagent. Aqua regia. This is only important to dissolve gold or silver powder. Dissolution. This is the best method to confirm Ti. After cooling dilute with 5 x water and add several drops of 3•o H20•.. A specific yellow-orange coloration occurs. Metal analysis. This will give information for talc or mica. Microscope. Mica platelets will show beautiful inter- ference colours under the microscope. Compare with reference substances. Market survey of eye make-up. Analytical results Sampling from the market was done in January 1972. There were nine brands with 111 samples. All colour tints of three brands were taken. Of the other brands the brown, grey and black colours were omitted, because they were chemically less interesting. All kinds of products were sampled: liquid liner, pencil liner, shadow liquid, shadow compact powder, shadow stick, shadow ointment, mascara wax cake, mascara liquid. Two unidentifiable samples were found in the 111 samples--one blue, one violet. There were 33 GREEN eye make-up preparations 28 contain chromic oxides 7 contain iron oxides 2 contain ultramarines 3 contain aluminum powder
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)