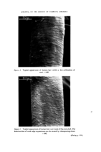
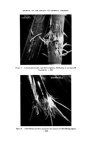
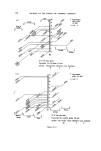
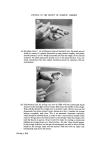
RISK-BEARING SUBSTANCES IN COSMETICS 195 o o o o o o 8 X X X X X • Sunscreenspots (Absorption meXo et .300 nm) E=I.O .... E_--/'O _ _ -. __ •_o.5 2 _ 280 :300 280 :300 280 300 280 300 280 300 nm nm nm nm nm Figure 9. Detection of a sunscreen spot on a tic plate. Scrape off the spot from the glass plate. Extract with 3 ml of methanol. Filter. Determine the uv spectrum of the solution in a 1 ml quartz cell, between 250 and 320 nm. If at this low concentration level (approx. 10 ppm) an appreciable absorption occurs at 300 nm, the tlc spot is a sunscreen. Examples: tlc of the samples no. 37, 35, 21, 24, 25 uv spectra of the spots. Solvent system' Diisopropylether-n. Hexane-Acetic acid (20 + 80-t- 1 vol). Unsaturated tank. Spots' For the extracts or reference solutions 5-10 pl. Visualization' Uv 254 nm. Gas-liquid chromatography of sunscreens Gaschromatographic conditions. Column: 6}/o Apiezon L and 10•o KOH on Chromosorb W 60-80. 150 cm diam. 0.25 inch, copper. Temperature column: isotherm 230øC. Injection port: 240øC. Detector: 250øC (flame ionization). Carrier gas' Nitrogen. Extraction: as under tic. Reference solution: as under tic. Injection: •-1 [tl.
196 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS The same column is used as for the aromatic diamines. (See hair colourants.) Benzocaine gives a good response. Not all of the reference sunscreens gave a response, but that was unimportant, as glc matching was the main purpose. Chemical analysis of sunscreens Group reactions for p.aminobenzoic acid and esters, and salicylate esters were carried out. P.aminobenzoic acid/esters. Ehrlich reagent (1 •o dimethylaminobenzaldehyde in 10•o HC1) gives a strong Yellow/Orange colour. Application directly on the sample or as a spray on a tlc plate. N.B. Many other compounds which are non-uv absorbers with aromatic amine groups show a positive Ehrlich reaction, e.g. sulphonamides. The possibilities for sunscreens are: p.Aminobenzoic acid, ethyl p. Amino- benzoate (= benzocaine), glyceryl p.aminobenzoate. N.N.dimethyl p.amino- benzoic acid esters do not give a reaction. Salicylic acid esters. After saponification, the free salicylic acid will give a violet colour with 1 •o FeCla. Experimental. Boil 0.5 g sample with 5 ml 8•o ethanolic KOH under reflux from 30 min. Cool, dilute with 40 ml water. Neutralize to pH 5-7. Add 1 drop 15/o FeCla. A violet colour appears, which will persist after the addition of an equal volume of ethanol (phenol gives a violet colour that fades with ethanol). General problems By means of a simple tlc/glc matching many of the sunscreens could be identified. An example is given in Fig. 10. Not all the sunscreens could be identi- fied, mainly because the range of reference compounds was not sufficient. Benzocaine in sunscreens In several samples of two brands, the analytical data indicate the presence of benzocaine. This substance has sunscreen properties, but acts as a local anaes- thetic as well. The presence of benzocaine was confirmed by its ir spectrum and its glc response, after isolation by column chromatographic means. Identification of benzocaine (ethyl p.aminobenzoate). Two samples (1 and 48) gave a strong yellow/orange coloration with Ehrlich reagent. Such a positive reaction, however, might also be given, not only by benzocaine, but also by the free p.aminobenzoic acid or its glyceryl ester, both of which are known sunscreens. Tlc separation of these three substances was achieved with the following solvent system:
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)