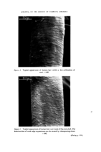
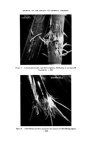
RISK-BEARING SUBSTANCES IN COSMETICS 187 Internal standard solution: 0.1 • in ethylacetate of 2CPFD. Ref. 25TDA solution: 0.15•o in ethylacetate. Standard curve: Prepare standard mixture by successive mixing of (internal standard solution) + (Ref. 25TDA solution) + (ethylacetate): 5-1-4, 5-2-3, 5-3-2, 5-4-1. These standard mixtures contain respectively per I•1: 0.150 0.300 0.450 0.600 pg 25TDA. Extraction: Weigh m g of the sample, which must contain 1-6 mg 25TDA. (For quantitative work this extract must be used within 1 h.) Mix this in a 10 ml volumetric flask with 5 ml internal standard solution and sufficient ethylacetate to a volume of 10 mi. Homogenize if necessary by heating on a waterbath. Add 1 g of anhydrous sodium sulphate, mix vigorously. Carry out the glc according to the projected injection sequence. Calculate the peak height ratio 25TDA/2CPFD. Find the equivalent pg 25TDA from the standard curve (a pg 25TDA). The •o 25TDA is (a/m) •o. height 25TDA height 2CPFD (A) 0.61 1.18 1.81 2.41 pg 25TDA per pl A/B 0.156 3.88 0.312 3.78 0.468 3.87 0.624 3.87 Mean A/B 3.85 Variation coeff. 1.3• The reliability of this method for our uncleaned extracts, however, is not better than 5•o, due to the retardance of the solvent peak to the baseline, which is caused by the impurities in the extract. Gas-liquid chromatography--phenols. The polyphenols can be separated, after silylation, on an OV 210 column (50•o Trifluoropropyl methyl silicone). In contrast to the aromatic diamines, crude extracts cannot be used in the glc pro- cedure, because many components of the basic support are silylated and interfere with the glc procedure. It is therefore necessary to clean the extracts as described in the following procedure. The polyphenols are isolated from the aromatic diamines, the aminophenols and also from impurities of the creme--or liquid-base of the hair colourant. The l•urified l•henol fraction is then silylated and separated on silicone column purified phenol fraction is then silylated and separated on silicone column (OV- 210). Satisfactory resolution is achieved between resorcinol and hydroquinone.
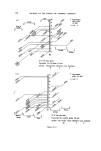
188 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 2'0 O' I0 0 '.'50 0-50 /zg 25 TDA per/zL Figure 6. Estimation of 2.5. toluene diamine by glc. Experimental Shake in a 250 ml separatory funnel: (4 g sample 5- 6 ml NaOH 1M 5- ca 500 mg Na-ascorbate to prevent oxidation 5- 100 ml dichloromethane). Shake vigorously. Wait at least 30 min for the separation of the phases. Discard the dichloromethane fraction. Acidify the aqueous fraction with 0.6 ml 35•o HC1. Extract with 50 ml, 25 ml, 25 ml dichloromethane successively. Dry the dichloro- methane fractions with Na-sulphate exsicc. Evaporate the clear extract on a waterbath until dry. Proceed with the silylation procedure in a small (approx. 5-10 ml) vial which can be closed tightly. Dissolve the dried residue of the extract in 4 ml ethylacetate and add successively: 0.2 ml Hexamethyldisilazane (HMDS) and 0.1 ml Trimethyl- chlorosilane (TMCS). Close the vessel and heat on a waterbath of 60øC for 5 min. Cool. The mixture is ready for glc. Glc-conditions: Column length 150 cm, ¬ inch, filled with 10•o OV-210 on Chromosorb WHP 80/100, glass. Column temperature 130øC isotherm. FID detector, temp. 230øC. Injectionport: 210øC. Carrier gas: Nitrogen 0.75 atm. Hydrogen: 30 ml/min. Air 300 ml/min.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)