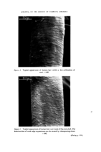
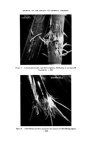
RISK-BEARING SUBSTANCES IN COSMETICS 193 body, by old and by young people, and intensively during several weeks annually. At that time the skin is exposed to the sun, the air and the water. It is possible that under such rigorous conditions of use the toxicological hazards of the active components will increase. To get an idea of the diversity in the use of the sunscreens in particular, analytical data of some 66 suntan preparations (24 brands) was collected. The identification was made mainly by tlc-glc matching with industrial sunscreens as references, and combined with chemical group reactions of two important classes of sunscreens. By the kind cooperation of the chemical industry, some 27 sun- screens were collected for reference analysis (Table VI). This number, however, is far below the possibilities mentioned in literature (22, 23) (Table VII). The list considered is given in Tables VI and VII. Table VI. List of reference industrial sunscreens Firmenich Solprotex 1, 2, 3 Givaudan Parsol mcx Parsol ultra Givtan F Laserson/Sabety Ecranosol Solecran Felton Sunarome GAF Uvinul ms40 Uvinul n539 Uvinul 400 Norda/Schimmel Angstrol Filtrosol A, B, Triple Naarden Solisoline A, B Merck Eusolex 3573 Eusolex 4360 Eusolex 161 Eusolex 232 Rhone-Poulenc Rhoditan L Dragoco Prosolal S9 Merck Eusolex 6653 Salicylates. 2.ethylhexyl p.methoxycinnamate. Mixture, not known. 2.ethoxyethyl p.methoxycinnamate. Salicylates. Unknown. 2.ethylhexylsalicylate. 2.hydroxy.4.methoxy.5.sulphonic acid. benzo- phenone. 2.ethylhexy1.2X.cyano.3.3 X.diphenylacrylate. 2,4 x.dihydroxybenzophenone. Salicylates. Salicylates. Unknowil. 4.phenyl.2.carbonicacid isoctyl ester.benzo- phenone. Unknown. 3.4.dimethoxyphenyl-glyoxylate Na. 2.phenylbenzimidazoline. Salicylates. Mixture of phenylacrylic and oxybenzoic esters. Dibenzalazin. Thin-layer chromatography of sunscreens In the tlc system used, several dark spots from a single extract usually appeared under uv light. To detect a sunscreen spot the following procedure was used. The spot is scraped off, extracted with 3 ml of methanol, filtered, and the uv
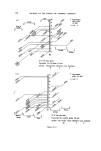
194 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Table VII. Inventory of sunscreens mentioned in the literature (22, 23) A. Para amino benzoic acid and esters: ethyl, butyl, propyl, glyceryl. B. N.N.Dimethyl para amino benzoic acid ester: isoamyl. C. Ortho aminobenzoic esters (anthranilic acid esters): methyl, menthyl, phenyl, benzyl, linalyl, cyclohexenyl, bornyl, isobornyl. D. Salicylic acid esters: menthyl, homomenthyl, phenyl, benzyl, glyceryl, dipropylene glycol, 2.ethylhexyl. E. Cinnamic acid esters and derivatives: 2.ethylhexyl p.methoxy cinnamate, benzyl cinnamate, 2.ethoxy ethyl p.methoxy cinnamate, menthyl cinnamate, benzyl p.methoxycinnamate, i.butyl salicyl cinnamate, propyl p.methoxycinnamate, butyl cinnamoyl pyruvate, ct-phenyl cinnamonitrile, 2.ethylhexyl 2X.cyano 3.3X.diphenylacrylate. F. Dihydroxycinnamic derivatives: umbelliferone, methylumbelliferone, methyl aceto-umbel- liferone. G. Trihydroxycinnamic acid derivatives: esculetin, I].methyl esculetin, dafnetin, glycosides of esculin and dafnin. H. Benzophenone derivatives: 2.4X.dihydroxybenzophenone, 4.phenylbenzophenone, carbonic acid isooctylester. 1. Quinine oleate, tannate, stearate, bisulphate. J. Coumarin derivatives: 7.hydroxy-, 7.methyl-, 3.phenyl. K. Digalloyltrioleate. L. Dibenzalazine. M. Dibenzalacetophenone, benzalacetone. N. Benzimidazolen, phenylbenzimidazolon sulphonic acid Na. O. 2.Phenyl benzoxazole, methylnaphthoxazole. P. 2.Phenyl benzothiazole. Q. Stilbene. R. 2.Acetyl bromo indazole. S. 8.Hydroxy quinoline, 2.phenyl quinoline. T. Butyl carbityl 6Lpropyl piperonyl ether. absorption spectrum determined between 250 and 320 nm. At such a low con- centration level (approx. 30 gg per 3 ml = 10 ppm) a sunscreen still has an absorption maximum at near 300 nm. Other dark spots of the chromatogram, such as from preservatives, will not have an appreciable absorption at 300 nm (see Fig. 9). Experimental Extraction: Dissolve 1 g of the sample in 4 ml methanol. Heat gently. Add 1 g Na2SO4 exsicc. Mix thoroughly. Use upper methanol layer for tlc or glc. Reference solution: Use 1 • w/v solutions of reference sunscreens in methanol. If the sunscreen is not soluble in methanol, dissolve by adding small amounts of water to the mixture until a clear solution is obtained. Tic-plates: Glass plates covered with a 0.25 mm layer of silica GF (with fluorescent indicator), and activated at 105øC for « h.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)