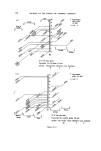
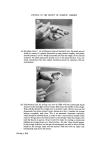
RISK-BEARING SUBSTANCES IN COSMETICS 189 Example of retention times: Pyrocatechol: 2.3 min Nitrobenzene: 2.9 min Resorcinol: 3.7 min Hydroquinone: 4.1 min Pyrogallol: 5.1 min Phloroglucinol: 10.7 min a-Naphtol: 12 min b-Naphtol: 13 min. See Fig. 7. Quantitative Estimations Quantitative determinations of resorcinol and hydroquinone are possible with nitrobenzene as an internal standard. Standard curve: Silylate 4 mixtures: each containing 2 ml of 0.25•o v/v nitro- benzene and 2 ml of a solution in ethylacetate of successively: 0.6--0.9--1.2--1.5 mg Resorcinol and the same amounts of hydroquinone. Inject 2 [tl for glc. 2O 4O % 5o 9C 0 I Figure 7. 2 3 4 5 I0 Min Glc separation of a silylated reference mixture of several poly- phenols used in oxydative hair colourants.
190 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Determination: The amount of Resorcinol (Res) or Hydroquinone (HCH) in the sample should be 0.4-1.6 mg. Prepare the purified extract as described under 'Experimental'. Dissolve the residue in 2 ml ethylacetate. Add 2 ml of the solution of the internal standard and silylate. Use 1-2 •1 for glc injection. Calculation: Find the peak height ratio RES/Nitrobenzene or HCH/Nitro- benzene. Find the corresponding mg RES or HCH in the standard curve. As the weight of the sample is known, the • can be calculated. Recoveries of 94• of the RES in experimental samples was achieved. Difficulties with chlororesorcinol and hydroquinone. In one sample a peak appeared on the hydroquinone place, after the resorcinol peak. The separation, however, was better than usual. The recorder reached the baseline, which was not the case in the standard chromatograms. The column temperature was lowered from 130øC to 100øC, a mixture of silylated hydroquinone and the extract was injected. Three peaks were deafly separated: resorcinol-hydroquinone and the quasi hydroquinone peak. Further trials with reference compounds, and tlc information led to the identification of 2.Chlororesorcinol. See Fig. 8. Analytical results--oxidative hair colour intermediates The results of the identification of our samples are tabulated below. Sur- prisingly p.phenylenediamine was absent. The most important components are still 2.5.toluylenediamine, the three isomeric aminophenols, resorcinol, naphthol and 2 nitro.p.phenylenediamine. Brand A (12 samples): 11 with 25TDA 12 with 3AF 12 with RES 2 with 2NPFD 1 with 4NOFD Brand C (5 samples): 4 with 25TDA 3 with 3AF 3 with RES 1 with 2NPFD Brand E (8 samples): 5 with 25TDA 2 with 2AF 3 with 4AF 3 with 2NPFD 8 with RES Brand B (9 samples): 7 with 25TDA 4 with 3AF 5 with RES 4 with 2NPFD Brand D (7 samples): 5 with 25TDA 2 with 3AF 3 with RES 3 with 2NPFD Brand F (11 samples): 10 with 25TDA 1 with 2AF 3 with 3AF 4 with 4AF 5 with RES 1 with AN
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)