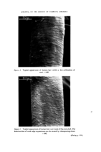
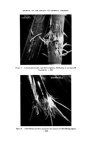
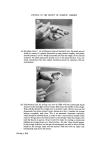
184 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS the first direction of the 2-dimensional procedure. Visualization is by iodine vapour. Determine the optimal amount of the extract and use twice this amount for the 20 x 20 cm tlc plates. Two-dimensional tlc. The spots on the two-dimensional tlc plate are identified by relative positions to the standard-mixture pattern. This is shown on Fig. 4. Colouring observation in the iodine tank will give some aid. It is important to remove all residual acetic acid of the second solvent from the plate (by heating for 10 min approx. at 50øC) in order to achieve optimal colouring. The conditions used were the following. Silica H plates (self-made) 20 x 20 cm, 0.25 mm thick, activated 1 h at 105øC. First direction: (unsaturated tank)Acetone- chloroform-toluene 35-25-40 vol. 1 h. Dry 15 min. Second direction: (saturated tank) 1 h 45 min. Chloroform Acetic acid-H20 50-45-5. Dry 10 min at 50øC, until smell of acetic acid has disappeared. Visualization: Put in empty chromatographic tank with several crystals of iodine. Observe starting and definite colours. Leave « h in tank. Take it out. Excess iodine will disappear, leaving the definite colours of the spots. PFD starts green, change to violet/brown MFD yellow/brown 25TDA starts green, change to violet/brown 24TDA yellow/brown 24DAF violet/brown 24DAA yellow/brown M4AF violet/brown AN violet BN yellow RES grey/brown HCH yellow PGL brown 2NPFD red 4NOFD orange 2AF yellow/brown 3AF brown 4AF violet/red. Optimal results depend on the quality of the tlc plates. We have had poor resolution with several kinds of precoated plates, as well as with self-made plates with gypsum binder, as well as for colour display by iodine vapour, and the shape of spots. Gas-liquid chromatography--amines. Doubts might still remain, especially with difficult pairs, such as resorcinol/hydroquinone, 2.5.toluylene diamine/2.4.di- aminoanisol/m. phenylenediamine, 2.4.toluylene diamine/4.aminophenol.
JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Figure 4. Two-dimensional resolution of a reference mixture. l, p.phenylene- diamine 2, 2.5.toluylenediamine 3, 4.aminofenol 4, 3.aminofenol 5, 2. nitro.p.phenylenediamine 6, 2.aminofenol 7, pyrogallol 8, 4.nitro.o. phenylenediamine 9, hydroquinone 10, resorcinol 11, b-naphthol 12, a- naphthol. (Facing p. 184)
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)