J. Soc. Cosmet. Chern. 27 289-300 (1976) ¸ 1976 Society of Cosmetic Chemists of Great Britain The chemistry of human hair cuticle•III: The isolation and amino acid analysis of various subfractions of the cuticle obtained by pronase and trypsin digestion J. A. SWIFT and B. BEWS* Synopsis--The course of digestion of human HAIR CUTICLE by PRONASE and TRYPSIN has been followed both gravimetrically and by the ELECTRON MICROSCOPE examination of digested hair sections. This has permitted the identification of three subfractions of the hair endocuticle. The significance of the AMINO ACID analyses is discussed. INTRODUCTION In the first two papers of this series we described a new technique for the physical isolation of cuticle from human hair (1) and the enzymatic isolation of the A-layer and cell membrane complex from the cuticle (2). Our attention has now turned to an examination of those cuticular components dissolved by the proteo~ lytic enzymes trypsin and pronase. Although Bradbury and Ley (3) have shown already that pronase can be used for the isolation of whole endocuticle from wool, the present experiments are more comprehensive and have enabled us to * Unilever Research Isleworth Laboratory, 455 London Road, Isleworth, Middlesex. 289
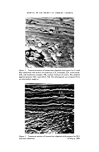
290 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS identify and isolate for analysis new and interesting subcomponents of the endo- cuticle of human hair. METHODS AND MATERIALS The experimental procedures were broadly the same as those described in the previous paper (2), namely (a) a study of the gravimetric course of digestion of isolated cuticle by pronase and trypsin, (b) the electron microscope examination of human hair sections after digestion with the enzymes to establish which mor- phological components are dissolved, and (c) the amino acid analysis of various identifiable fractions isolated from the bulk cuticle. The enzyme solutions used were 0.1•o w/v pronase (Calbiochem grade B) and 0.1 •o w/v trypsin (Sigma, 2 x crystallized) in 0.1M ammonium acetate/am- monia buffer at pH 7.8 containing 10•o w/v ethanol (the ethanol was used to minimize bacterial growth and also to facilitate the wetting of the samples to be treated (3)). The corresponding buffer at pH 7.8 without the added enzymes was used for washing the various insoluble fractions and hair sections. The insoluble fractions were obtained as described earlier (2) and the soluble fractions by evaporation in vacuo to small bulk followed by freeze drying. The heavy metal stains used for treating the enzyme-digested hair sections were different from those used in the previous paper. They were (a) 0.1N silver nitrate to which 0.880 ammonia was carefully added until the precipitate just dissolved grid-mounted sections were treated for 30 m at room temperature, followed by 2 x 15 m immersions in distilled water and then dried, and (b) 10•o w/v dodecatungstophosphoric acid (PTA) (BDH Analar grade) in 50•o w/v ethanol filtered through 0.45 [tm Millipore membrane grid-mounted sections were treated for 2 h at 40øC followed by a 20-s rinse in 50•o ethanol and rapid drying of the grid on the corner of a filter paper. RESULTS Course of digestion of isolated cuticle with trypsin and pronase A graph showing the loss in weight of hair cuticle against time of digestion with trypsin and pronase is shown in Figure 1. The rate of digestion with trypsin was fairly rapid for the first 5 h and then the rate slowed down so that between 10 and 60 h it was about 1/12 of the initial rate. No further digestion occurred after 70 h by which time 20•o of the cuticle had dissolved. It is clear that the two dis- tinguishable rates of digestion arise through the dissolution of two separate com- ponents of the cuticle. By extrapolating the linear portion of the curve between 10 and 60 h, it was established that the fast-dissolving component constitutes
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)