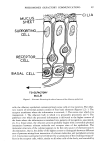
58 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS (60) (61) (62) (63) (64) (65) M. G. Kalogerakis, Psychosom. Med., 25,420 (1963). D. M. Zahorik and R. E. Johnson,J. Comp. PhysioL Psychol., 90, 57 (1976). H. Wiener, N.Y. StateJ. Med., 66, 3153 (1966). A. Comfort, "The Joy of Sex," Crown, New York, 1972. M. Rhodes (Producer), London, BBC Horizons/Nova, 1972, film made for television. M. E. Seligman and J. L. Hager, eds., "Biological Boundaries of Learning," Appleton--Century-- Crofts, New York, 1972. (66) N. Tinbergen, "The Study of Instinct," Oxford, Clarendon Press, 1951. (67) P. Rozin and J. W. Kalat, "Biological Boundaries of Learning," M. E. Seligman andJ. L. Hager, eds., Appelton--Century--Crofts, New York, 1972. (68) J. Garcia, F. R. Ervin, C. H. Yorke, and R. A. Koelling, Science, 155,716 (1967). (69) I. P. Pavlov, "Lectures on Conditioned Reflexes," International Publishers, New York, 1928. (70) B. F. Skinner, "Psychology--A Study of a Science," S. Koch, ed., McGraw-Hill, New York, 1959. (71) R. B. Lockard, Am. Psychol., 25,168 (1971). (72) H. Ellis, "Studies in the Psychology of Sex," Vol. I, Random House, New York, 1942.
J. Soc. Cosmet. Chem., 29, 59-64 (February 1978) Application of lower titanium oxide in cosmetics FUKUJI SUZUKI, SHOJI FUKUSHIMA, TAKEO MITSUI and SABURO OHTA Shiseido Laboratories, Nippa-Cho, Kohuku-Ku, Yokohama, Japan 223 Received March 7, 1977. Presented at Annual Meeting, Society of Cosmetic Chemists, December 7, 1976, New York. Synopsis Carbon black and iron black (FeaO4) are black pigments commonly used in the cosmetic industry. Due to its hydrophobic character, carbon black has poor dispersibility in water, and iron black, due to its ferro-mag- netic character, is not readily dispersible in any liquid. In order to solve these problems the authors investi- gated the possibility of synthesizing LOWER TITANIUM OXIDES and using them as a black pigment IN COSMETICS. The general formula of the lower titanium oxide is TinO•n-• in which n can be any positive in- teger. When n is small, the resultant compound is bluish-black. As n increases, the compound becomes gray. The lower titanium oxide most suitable for cosmetics was obtained by calcining a mixture of a titanium dioxide pigment and a metallic titanium powder in a vacuum electric furnace. While it has a tinting strength corresponding to that of carbon black and iron oxide black, it is superior in many other respects when used in cosmetics. INTRODUCTION Black pigments, commonly used in the cosmetic industry at the present time, are two in number: namely, carbon black and iron oxide black (FeaO4). However, both are pig- ments not easily formulated into make-up cosmetics of the dispersion type. Carbon black has a hydrophobic surface-and its surface area is extremely large (about ten times that of titanium dioxide pigment). Iron oxide black, though considered to have a hy- drophilic surface, tends to strongly coagulate in any medium due to its ferromagnetic character. This gives rise to poor dispersibility with considerable viscosity changes when the formulation calls for producing different shades of cosmetics and, further- more, color separation occurs on the surface of the cosmetics when this material is used along with other pigments such as titanium dioxide. In order to solve these problems, we explored many black pigments showing greater dispersibility and most obviously with a high safety factor when used in cosmetics. In the lower titanium oxides, we noted several pigments with great possibilities. It is well known that there are the three crystal forms of titanium dioxide: anatase, rutile and brookite. In the cosmetic or paint industry, pigments of the former two types are used. Titanium dioxide is a very stable compound. On the other hand, titanium can 59
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)