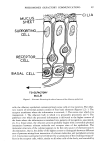
60 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS make various oxides which are expressed as TinO2n_• in which n can be any positive in- teger. They are generally called lower titanium oxides (1-4). Ehrlich describes two methods for their synthesis. The first involves the reduction of titanium dioxide (Ti2Oa or TiaO5 is synthesized as a result of the loss of oxygen when TiO2 is heated in a hy- drogen stream at temperatures of 1200 to 1500øC). The second occurs as a phase reac- tion by heating a mixture of titanium dioxide and metallic titanium (in appropriate ratio) in a vacuum. Ehrlich referred to the fact that the lower titanium oxide has a dark shade. We investigated the lower titanium oxides in detail and found that, as expected, some of them had superior qualities compared to the above two black pigments. EXPERIMENTAL MATERIALS A metallic titanium and three kinds of titanium dioxide pigments were used. The tita- nium powder was an analytical grade reagent (above 99 per cent) from Kishida Chemical Co. Ltd., of which the specific BET surface area (SN•) was 0.24 m•/g. The tita- nium dioxides (TiO2) were all commercially available powders for pigment use of which SN2 was 8.5 (#328, NL Industries), 25.0 (P-110, Degussa) and 54.0 m•/g (P-25, Degussa), respectively. All the materials used in the experiment were free of any heavy metals which are prohibited from use in cosmetics. METHOD OF PREPARATION The titanium and titanium dioxide were mixed in predetermined ratios using a ball-mill or a portable Henshel mixer. About 70 g of the mixture was heated in a vacuum electric furnace at 600, 800, and 1000øC at a vacuum of below 10 -2 torr for 1, 2, 4, 8, and 20 hr. Figure 1 illustrates the arrangement of the equipment used for the calcina- tion process. After heating, the furnace was allowed to. cool to below 200øC by turning off the electric current while still maintaining the vacubm then the product was taken out. Above this temperature, the lower titanium oxide is apt to oxidize and become white. The product was pulverized in a mortar since it aggregated slightly by sintering. --- - -._ Temperature (q) %% ..... •olle• Dry,,2g r I[• qiartz5 i• •xFx•_• • Tubular Electric Furnace • • •c•od • Gau• Rotary Pump Figure 1. Arrangement of equipment used in the calcination
LOWER TITANIUM OXIDE 61 METHODS FOR TEST The following procedures were carried out in order to characterize the synthesized ma- terials: a) X-Ray diffraction.' Crystallographic assessment of the products was carried out using a Rigaku Denki X-ray diffractometer (RU-3) by Cu-K• radiation. b) Color measurement.' A CC-1 Color Computer (Tokyo Shibaura Electrics), a visible light absorption spectrometer equipped with a minicomputer, measured the color of the products and that of 10 per cent mixtures in TiO• # 328. The latter is for assessing the tinting strength of the products. The L-value in the Hunter's equation, which expresses lightness, will be used for referring to the blackness and tinting strength of the products. Other chromatic values such as hue (= b/a) and saturation [ = (a • + b 2)•/2 ] were of no use since the products were achromatic. c) Viscosity and superficial appearance of a slurry.' Products were mixed with TiO• # 328 at the ratios 0/100, 15/85, 25/75, 50/50, 75/25, and •00/0 using an Emide mixer. The mixture (70 g) was dispersed into a tetradecane-sorbitan monooleate 34/4 v/v solution, which was passed through a three-roller mill. Viscosity of the slurries was measured with a Ferranti-Shirley cone-plate viscometer under the condition to give a maximum shear rate of 1720 sec -• with a 10 sec sweep time. Apparent viscosity was obtained from the shear stress at the maximum shear rate. Attention was paid to the appearance of the slurry surface throughout this experiment. RESULTS AND DISCUSSION OPTIMUM CONDITION FOR SYNTHESIS In order to determine the shortest time required for the calcination reaction to be com- pleted, the existence of unreacted starting materials such as TiO2 and Ti in the product was examined by X-ray diffraction. Figure 2 shows an example of the results in which one can note the progress of the reaction. With time, the diffraction peaks of the start- ing materials decreased in intensity and the intensity of the end product increased. Figure 2 indicated that the reaction had been completed after 4 hr. Table I summarizes the time required for the completion of the reaction at different temperatures. Table I Heot T T,ton,um Treolment After Ihr a! 800C 2hrs al T 3 P3 T,•O• 800C R 2 2 n Afte• p3 • p2 20 30 40 Bragg's Angle (2•), Degree Figure 2. An example of the change in X-ray diffraction patterns with time for a Ti - TiO2 mixture
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)