J. Soc. Cosmet. Chem., 40, 75-90 (March/April 1989) A simple method to avoid sandiness and enhance deodorant efficacy of soaps containinD TCC G. SAUERMANN, U. HOPPE, M.-C. LENEVEU, M. R•CKL, O. STELLING, Beiersdorf AG, Unnastrasse 48, D-2000, Hamburg 20, Federal Republic of Germany. Received October 12, 1988. Synopsis The "sandiness" of soap can be caused by TCC. This is shown in that the TCC-content of "sand" grains is very high compared to its microenvironment within the soap and by EDXA-analysis of C1. The bioavail- ability of TCC of "sandy" soap (sandiness is caused by agglomerated TCC) is considerably lower than that of regular smooth soap. This was shown by analytical determination by HPLC of the TCC-level at the skin surface. Solubilization of TCC by means of nonylphenolether enhances the transfer of TCC. The amounts of TCC deposited on the skin surface while using soaps containing 1.3% powdered or 0.26% solubilized TCC are comparable. The amount of TCC transferred to the skin surface is approximately linearly proportional to the content of soap of TCC. Addition of wool wax alcohols as refattening agents increases the bioavailability of TCC. An excess of solubilizer decreases the transfer rate of TCC. Sniff tests prove the equivalence of deodorant soaps containing either 0.26% solubilized or 1.3% powdered TCC. INTRODUCTION This publication presents a simple method to avoid sandiness and to increase the biolog- ical availability of TCC, i.e., the amount of germicide that can be transferred from the soap-water mixture to the skin surface during normal use. The basic method used to avoid a gritty surface structure is to solubilize TCC and to add the mixture to the soap base in amounts necessary for effective deodorant action. MATERIALS AND METHODS PREPARATION OF THE SOAP CAKE 0.5 kg of TCC was solubilized in 3.0 kg of nonylphenolpolyglycol ether (Marlophen 814, Chemische Werke, HiSis, FRG) having an average degree of ethoxylation of 14 (1). This mixture was added in portions to 100 kg of soap base in noodle form, together with perfume and 1.65 kg of color paste which may contain 0.6 kg of wool wax alcohols as well as other ingredients. After thorough mixing, the homogenized mixture was processed in the usual manner and finally pressed. COMPOSITION OF THE SOAP CAKES TO BE TESTED Soap cakes prepared in the manner described above (with different contents of TCC and wool wax alcohols) were examined: 75
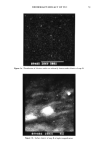
76 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS A. Containing 1.3% regular TCC (standard version, commercially available) with no sandy texture. B. Containing 1.3% regular TCC (standard version, commercially available), gritty surface when being used. C. Containing 0.13% solubilized TCC. D. Containing 0.26% solubilized TCC. E. Containing 0.5% solubilized TCC. F. Containing 1.3% solubilized TCC. G. Containing 0.26% solubilized TCC + 0.06% wool wax alcohols. H. Containing neither TCC nor wool wax alcohols. I. Soap G + 0.5 % dimethylpolyglycol ether. K. Soap D + 5.26% nonylphenolpolyglycol ether additionally. Soaps A and B were prepared on a big scale in the factory soaps C-K were prepared on a small scale in the laboratory. EDXA-- INVESTIGATION The surfaces of pieces of soaps (B, C, F) were freshly cut with a knife. The surfaces-- covered with carbon--were investigated by a combined SEM (scanning electron micro- scope, Hitachi S-500) and EDXA (X-ray-fluorescence, Kevex • X System 7000) tech- nique. The distribution of chlorine was determined to visualize the content and the tendency of TCC to form aggregates. Sodium was determined to show the homogeneity of the distribution of Na-stearate/palmitate. QUANTITATIVE DETERMINATION OF TCC TCC, dissolved in ethanol, was determined by HPLC (Hewlett-Packard 1084 B, column Lichrosorb RP8 (10 •), solvent H20:ethanol = 23:77, injection volume 10 •1, wavelength 266 nm). SAMPLING PROCEDURE FOR DETERMINATION OF THE DENSITY OF TCC AT THE SKIN SURFACE The quantity of TCC/area unit of skin surface was determined in the following manner: The skin area (the inner side of the lower forearm), previously washed with soap, was covered with a funnel (0 60 mm) 5 ml of ethanol were poured into the funnel the skin site, funnel, and solvent were shaken continuously for two minutes then the solvent was transferred to a glass bottle, which was sealed afterwards by means of a septum. DETERMINATION OF TCC BIOAVAILABILITY OF SOAPS A AND B The comparison of "sandy" soap (B) with soap (A) of good quality was done as follows: ten female volunteers used running tap water of 37øC to wash one lower forearm with one of the two soaps. Analyses of TCC were performed as described above. SKIN SURFACE CONTENT OF TCC IMMEDIATELY AND 12 HOURS AFTER USE OF SOAPS To find the equivalence level between powdered and solubilized TCC, the quantitites of
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)