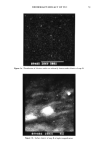
DEODORANT EFFICACY OF TCC 81 Figure 3a. Distribution of chlorine within an arbitrarily chosen surface district of soap C. .( ' • '.• •-• I • IIi-•'I •11f[I ', • I l ' i I NUCLEAR CHAROE i '.--J I ............... • • • _, _1 .... !•._i_ ' _. Figure 3b. Relative quantities of elements within the surface of soap C (investigated surface area identical with that of Figure 3a).
82 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS analysis of chlorine. Figures 2b and 3b show the same integral intensities of Na and S in the surface districts the density of chlorine-indicating spots in Figures 2a and 3a are in accordance with the content of chlorine. BIOAVAILABILITY OF TCC FROM DIFFERENT SOAPS The velocity of heterogeneous reactions is normally influenced by the distribution of the reaction partners, i.e., the rate of transfer of TCC from the soap to the skin surface will probably depend on such factors as the distribution of the deodorant, the duration of interaction, the reaction temperature, and other ingredients of the soap, which might have a promoting or interfering influence. Presumably the quantity of TCC on the skin surface should strongly influence the deodorant effect. SOLUBILIZATION ENHANCES TCC TRANSFER The logical consequence of these results was to look for the effect of TCC dispersed in micelles or evenly dissolved in soap as individual molecules. Some preliminary studies showed that soaps (e.g., soap F) containing solubilized TCC deposited much more TCC on the skin surface than soaps containing powdered TCC (e.g., soap A). The density of TCC on the skin surface was 0.24 (•g/cm 2) using soap A and less than 0.1 (•g/cm 2) using soap B (Figure 4). One might speculate that soaps with a gritty texture due to TCC will probably show a much less pronounced deodorant effect than soaps with the same content of TCC but with fairly evenly distributed powdered TCC. These interesting but preliminary results were confirmed by the following. DETERMINATION OF TCC BIOAVAILABILITY WITH REGULAR USE OF SOAPS OF DIFFERENT TCC CONTENT Figure 5 shows the relationship between transferred TCC and TCC content of the soaps: a) The rate of TCC transfer is an approximately linear function of the TCC content of the soap used. b) There is a slight tendency to higher values with longer test periods. c) After solubilization the TCC content of the soap can be reduced to less than ¬ of the TCC content of soap A containing 1.3% powdered TCC to reach the same level of TCC at the skin surface (1). Statistical differences were found between C and all other tested soaps, between D and E, D and F. No differences were found among A, B, and E. INFLUENCE OF THE DURATION OF WASHING AND OF REFATTENING AGENTS The influence of the washing period on the quantity of transferred TCC is summarized in Figure 6. After 10 and 120 seconds, significantly higher values (95%) were found on the arms treated with soap G. This may be due to a decrease of "free" solubilizer in soaps containing refattening agents. CORRELATION BETWEEN THE LEVEL OF SKIN SURFACE LIPIDS, SKIN SURFACE ROUGHNESS, AND TCC TRANSFER There is a weak correlation between the content of skin surface lipids and the quantity
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)