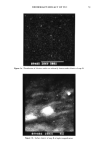
124 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 6 2 o - // - _ ///(3 0 12 24 36 48 60 72 Time (h) Figure 4. Penetration profile of vitamin E across hairless mouse skin. Key: (VI) HPLC assay. (O) Radioac- tivity counting ( X 1/10). m Calculated (k o = 0.0038 s- •, A = 5.6 X 10 -6 S- •). __ Calculated (k o = 0.0038 s- •, A = 0). - .... Calculated (X 1/10) (k 0 = 0, no metabolism). Diffusivity in stratum corneum = 8.4 X 10 -• cm2/s. Diffusivity in viable skin = 7.1 x 10 -8 cm2/s. Partition coefficient between stratum corneum and viable skin = 18.9. Concentration of the skin surface = 22.3 p•mol/ml. Thickness of stratum corneum = 10 p•m. Thickness of viable skin = 370 p•m. The numbers on the curves are the values of decay rate constant A. lated profile with the rate constant A of 5.6 x 10 -6 s-1 agrees fairly well with the experimental profile. It is also found that the enzyme activity in the hairless mouse skin, if freshly excised, remains unchanged during at least 24 hours after the onset of the in vitro skin penetration experiment. CONCLUSION The present study clearly indicates that the permeation of a radiolabeled compound appears to be significantly higher than the penetration of the actual compound due to skin bioconversion. It is therefore important, when evaluating percutaneous absorption of drugs, not only to determine the overall permeation rate but also to investigate whether or not enzymatic bioconversion occurs in the skin. ACKNOWLEDGMENT Radiolabeled vitamin E was supplied by Hoffmann-La Roche (Nutley, NJ) and their help is greatly appreciated.
SKIN PENETRATION 125 REFERENCES (1) (2) (3) (4) (5) (6) (7) (8) (9) (10) (11) (12) L. J. Machlin, Handbook of Vitamins (Marcel Dekker, New York, 1984). L. J. Machlin, Vitamin E, A Comprehensive Treatise (Marcel Dekker, New York, 1980). J. N. Counsell and D. H. Hornig, Vitamin C (Applied Science Publ., London and New Jersey, 1981). R. E. Keith, B. M. Chrisley, and J. A. Driskell, Dietary vitamin C supplementation and plasma vitamin E levels in humans, Am.J. Clin. Nutr., 33, 2394-2395 (1980). K. Tojo, Mathematical modeling of transdermal drug delivery, J. Chem. Eng. Japan, 20, 300-308 (1987). K. Tojo, J. A. Masi, and Y. W. Chien, Hydrodynamic characteristics of an in vitro drug permeation cell, I&EC Fundamentals, 24, 368-373 (1985). K. Tojo, "Design and Calibration of In Vitro Permeation Apparatus," in Transdermal Controlled Sys- temic Medications, Y. W. Chien, Ed. (Marcel Dekker, New York, 1987), pp. 127-158. K. Tojo and A. R. C. Lee, Bioconversion of a provitamin to vitamins C and E in skin, J. Soc. Cosmet. Chem., 38, 333-339 (1987). G. R. Buettner, A. G. Motten, R. D. Hall, and C. F. Chignell, ESR detection of endogenous ascot- bate free radical in mouse skin, Photochem. Photobiol. 46, 161-164 (1987). K. Tojo, C. C. Chiang, and Y. W. Chien, Drug permeation across the skin: Effect of penerrant hydrophilicity, J. Pharm. Sci., 76, 123-126 (1987). T. Shiratori, Uptake, storage and excretion of chylomicra-bound 3H-o•-tocopherol by the skin of the rat, Li• Sci., 14, 929-935 (1974). K. Tojo, K. H. Valia, and Y. W. Chien, Bioconversion of estradiol esters in hairless mouse skin in vitro, Biochem. Eng. J., 33, B63-B67 (1986).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)