338 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS EE is typically determined by separating free from encapsulated compound and assaying one or both fractions for the compound of interest. Various separation techniques have been described and compared in the literature. Current procedures utilize size exclusion gel filtration (6-12), centrifuged size exclusion gel mini-column (13-15), dialysis (16,17), ion exchange columns (18), ultracentrifugation (6,19-21), and discontinuous density gradients (22,23). For the encapsulation of hydrophilic molecules, typically, small-size vesicles (400 nm in diameter) encapsulating sucrose or carboxyfiuorescein have been evaluated (3,5,13,22,24). Inulin is a 5,000-Da hydrophilic molecule that has been reported to be localized in the skin following delivery from liposomes (1,25,26). We were interested in quantitating the EE of inulin in different liposome systems for i, vitro skin penetration experiments (27). Using a phosphatidylcholine (PC)/inulin liposome system, we tested the centri- fuged-column method, dialysis, and the discontinuous density gradient separation method. After selecting the latter as the optimum separation method for this particular system, we evaluated various encapsulation procedures and liposome compositions. MATERIALS AND METHODS CHEMICALS Soybean phosphatidylcholine (PC) and phosphatidylethanolamine (PE) were obtained from Avanti Polar Lipids (Alabaster, AL). Inulin, cholesterol, cholesteryl hemisuccinate (CHEMS), dicetylphosphate (DCP), oleic acid (OA), o•-tocopherol, N-2- hydroxyethylpiperazine-N'-2-ethanesulfonic acid (HEPES), and disodium EDTA were purchased from Sigma (St. Louis, MO). [Carboxyl-•4C]inulin (•4C-Carb-I, specific ac- tivity 2.05 mCi/g) was purchased from American Radiolabeled Chemicals (St Louis, MO), and L-3-phosphatidylcholine 1,2-di[1-•4C]palmitoyl (•4C-DPPC, specific activity 100-120 mCi/mmol) was obtained from Amersham (Arlington Heights, IL). Ficoll © 400-DL and Sephadex G-25 fine and G-50 fine were purchased from Pharmacia Biotech (Piscataway, NJ). The scintillation cocktail was Scintisafe Plus 50% (Fisher Scientific, Pittsburgh, PA). All other chemicals were of analytical grade. The buffer was HEPES buffer, pH 7.4 (20 mM HEPES, 150 mM NaC1, 0.1 mM EDTA). PREPARATION OF •4C-DPPC LIPOSOMES PC liposomes labeled with •4C-DPPC were prepared by the film method (28). Briefly, 30 mg PC, 1.65 pg •4C-DPPC (0.25 pCi), and 0.3 mg o•-tocopherol in chloroform solution were pipetted into a 100-ml round-bottom flask. The flask was mounted onto a rotary evaporator and the chloroform evaporated under a gentle N 2 stream. Once a dry lipid film was obtained, vacuum was applied for one hour to remove any traces of solvent. The film was then hydrated at room temperature for one hour with 5 ml buffer. The final PC concentration was 6 mg/ml, and the 14C-DPPC radiochemical concentra- tion was 0.05 pCi/ml. PREPARATION OF •4C-CARB-I LIPOSOMES Liposome preparations containing 2% w:v inulin spiked with •4C-Carb-I were prepared by four different methods. For the film method (28), 48 mg PC and 0.48 mg o•-to-
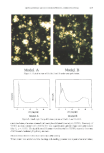
ENCAPSULATION INTO LIPOSOMES 339 copherol in chloroform solution were pipetted into a 25-mm x 175-mm test tube with a 24/40 ground-glass joint. The test tube was mounted onto a rotary evaporator, and a dry lipid film was obtained as described above. The film was then hydrated at room temperature for one hour with 2 ml buffer containing 2% w:v inulin and 0.14 pCi/ml •4C-Carb-I. The liposome preparation was submitted to five freeze/thaw cycles (isopro- panol-dry ice/25øC-water bath for 5 min). The final lipid concentration was 24 mg/ml. Three variations of the reverse phase evaporation method (RPE method) originally described by Szoka et M. (29) were studied. In protocols i and 2, 48 mg ofphospholipids, o•-tocopherol, and in some cases, other liposome wall components in chloroform solution were pipetted into a 25-mm x 175-mm test tube with a 24/40 ground-glass joint (22,24). The test tube was mounted onto a rotary evaporator and the chloroform evap- orated under a gentle N 2 stream. In protocol 1, once a dry lipid film was obtained, 6 ml of diethyl ether and 2 ml of buffer containing 2% w:v inulin and 0.14 pCi/ml •4C- Carb-I were added to the test tube. In protocol 2, 2 ml of diethyl ether and 0.67 ml of buffer containing 6% w:v inulin and 0.42 pCi/ml •4C-Carb-I were used. In both cases, the ether/buffer mixtures were sonicated for about 10-15s at 0-5øC to form a homo- geneous dispersion. Diethyl ether was then carefully removed under vacuum. In protocol 2, liposome preparations so obtained were then diluted 1:2 v:v with buffer, before use. The final liposome concentration was 24 mg/ml. For protocol 1, other concentrations were also studied. In the third RPE method (6), chloroform was used directly as the organic solvent instead of diethyl ether, and cholesterol and dicetylphosphate (DCP) were added as stabilizers. In a 100-ml round-bottom flask were dissolved 10.2 mg PC, 5.0 mg cholesterol, and 2.0 mg DCP, with 4 ml of chloroform. The flask was hand-shaken to dissolve all compo- nents, and 1 ml of buffer containing 2% w:v inulin and 0.14 pCi/ml 14C-Carb-I was added drop-wise. Chloroform was removed under vacuum. By this method, Alam et M. were able to encapsulate gelonin (a 30,000-Da protein) at a 77-79% efficiency (6). All liposome preparations were stored at 4øC under nitrogen before use. LIPOSOME SIZING Size distributions for PC and PC/Chol 9:1 mr liposomes prepared by RPE protocol 2 were determined using a HORIBA LA-900 laser scattering particle size analyzer (Horiba Instruments, Inc., Irvine, CA). The instrument combines QELS and Fraunhofer diffrac- tion to cover a range from -20 nm to -100 pm. Measurements were taken without sonication, and the refractive index was set at 1.19-0.00 i. CRYO-TRANSMISSION ELECTRON MICROSCOPY A PC liposome preparation prepared by RPE protocol 2 was examined by cryo- transmission electron microscopy (cryo-TEM) to characterize the microstructure. A 5-pl drop of liposomal dispersion was applied to a lacey carbon Formvar film-covered EM grid (Ted Pella, Inc., Redding, CA) in a controlled environmental vitrification system (30). Most of the solution was blotted from the grid with filter paper (Whatman •1) to form a thin film specimen, which was immediately vitrified by plunging into liquid ethane at its freezing point. The vitrified sample was examined at -172øC using a Gatan
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)