340 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS cold stage (Gatan, Inc., Warrendale, PA) in a Philips CM-12 TEM (Philips Electronic Instruments Co., Mahwah, NJ). Acceleration voltage was 100 kV. Micrographs were recorded on Kodak SO-163 film developed to maximum speed in full-strength Kodak D- 19 developer. CENTRIFUGED MINI-COLUMN SEPARATION METHOD The centrifuged mini-column method is a combination of Sephadex filtration and low- speed centrifugation, which prevents sample dilution typically observed in conventional size-exclusion gel filtration. It is non-destructive, fast (about 5 min/sample), and uses sample sizes in the 0.1-0.5 ml range. Free inulin is retained by the gel whereas lipo- somes are recovered in the eluate. The methods of Fry et al. (13) and Penefsky (15) were adapted as follows: Sephadex beads were allowed to swell in buffer. The barrel of a 3-cc syringe (Becton-Dickinson, Rutherford, NJ), fitted with drain disks (Corning Costar Corp., Boston, MA) was filled with Sephadex gel and inserted into a 15-ml conical centrifuge tube, supported at the top by the finger grips of the syringe. The whole assembly (syringe and tube) was centrifuged for two minutes at 100 g to remove excess buffer from the Sephadex beads. The syringe was transferred to a clean tube, and 0.5 ml of the test preparation was carefully applied to the Sephadex bead. The column was centrifuged as before and the eluate recovered for liquid scintillation analysis. Two grades of Sephadex, Sephadex G-50 fine and G-25 fine, were evaluated. Retention of free inulin by the Sephadex gel was tested using a solution of 2% w:v inulin and 0.14 laCi/ml •4C-Carb-I in buffer. Liposome recovery from the column was assessed with a •4C-DPPC liposome preparation (0.05 laCi/ml). Radioactivity levels in the eluates were determined by scintillation counting on a Beckman LS 7000 scintillation counter (Beck- man Instruments Inc., Irvine, CA). DIALYSIS The dialysis protocol described by Bangham et al. (16) was modified to account for the difference in diffusion rate between small ions and inulin. Dialysis tubing (Fisher Scientific, Pittsburgh, PA cut-off, 12,000 Da) was cleaned according to a standard procedure and rinsed with buffer. The lower end of the tube was tightly clipped, and 0.5-1.0 ml of the test solution was pipetted into the tube. The upper end was clipped tight and the sample dialyzed against 12-hour changes of 500 ml buffer. A solution of 2% w:v inulin and 0.14 laCi/ml •4C-Carb-I in buffer and •4C-DPPC liposomes was tested. Radioactivity levels in the dialysates and dialysis bag were determined by scin- tillation counting. DISCONTINUOUS FICOLL © DENSITY GRADIENT SEPARATION METHOD In the discontinuous density gradient method, layers (typically three or four in a 5-ml centrifuge tube) of decreasing density are carefully pipetted on top of each other, with the sample mixed in the layer of highest density. Upon centrifugation, separation occurs based on molecular weight. This method was used successfully by Betageri with a metrizamide gradient (31), by Heath et al. with a dextran gradient (23), and by Fraley
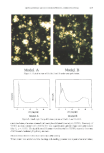
ENCAPSULATION INTO LIPOSOMES 341 et al. with a Ficoll © gradient (22). In our study, the protocol described by Fraley et al. (22) was adapted as follows: 0.6 ml of a 14C-labeled test preparation of known radio- chemical concentration was thoroughly mixed with 1.2 ml of Ficoll © 30%. One and one-half milliliters of the mixture was pipetted into a 5-ml polyallomer centrifuge tube (Beckman, Palo Alto, CA) and exactly weighed. Three milliliters of Ficoll © 10% were then carefully added on top of the first layer, followed by 0.5 ml of buffer. The tube was centrifuged at 100,000 x g for 30 min. Starting from the top of the centrifuge tube, 100- or 200-pl fractions of each layer were pipetted into scintillation vials and assayed for 14C by liquid scintillation counting. The presence of liposomes in these fractions was assessed visually. Fractions containing liposomes appeared milky, whereas non-liposome- containing fractions appeared clear. A solution containing 2% w:v inulin and 0.14 pCi/m114C-Carb-I in buffer was used to monitor the migration of inulin through Ficoll © upon ultracentrifugation. The migration of the liposomes was monitored separately using 14C-DPPC liposomes. Additionally, a solution containing 2% inulin, •4C-Carb-I, and empty PC liposomes (24 mg/ml) was also tested to ensure that there was no binding of inulin at the surface of the liposomes, which could result in an overestimation of EE. CALCULATION OF ENCAPSULATION EFFICIENCY (EE) EE is commonly defined as the number ratio of encapsulated molecules to total mol- ecules. To take into account variations that may simply arise from the amount of lipids used in liposomal preparations, some workers prefer to define EE as the amount of compound encapsulated per unit weight of lipids. In this study, we report both values. To calculate EE from Ficoll © separation data, the amounts of radioactivity (in CPM) recovered in each liposome fraction were pooled, and EE (in %) was determined using the following formula: EE (%) = 100' CPML/[AAs* m•] wherein CPM• is the total amount of radioactivity in liposome fractions (CPM), AA s is the average activity of the 30% Ficoll © layer before centrifugation (CPM/mg), and m• is the mass of the 30% Ficoll © layer in the centrifuge tube (mg). EE, expressed as mg inulin per mg lipid, was calculated as follows: EE (mg inulin/mg lipid) = lEE(%)* Cinulin]/CLipi D wherein Cinun n is the concentration of inulin (20 mg/ml) and CLiPi D is the concentration of phospholipid (variable, mg/ml). RESULTS AND DISCUSSION LIPOSOME CHARACTERIZATION Fresh PC and PC/Chol 9:1 mr liposomes prepared by RPE protocol 2 displayed similar size distributions, ranging from 2 to 20 lam (Figure 1). Analysis of a 21-day-old PC preparation showed a shift towards larger sizes (distribution between 2 and 100 lam). The shift for 15-day-old PC/Chol 9:1 mr liposomes was less significant (distribution between 3 and 30 lam). Such improvement in liposome long-term stability by addition of cholesterol was reported earlier (32). Vesicular structures displaying various mor-
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)