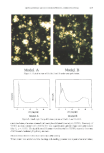
CROSS-ADAPTATION BY STRUCTURAL ANALOGS 371 Exposure to the E-isomer of EE3M21- -.•- E-EE3M2H - '- 10Z 1E 3M2H Adaptabort Recovery A 125 100 Exposure to the Z-isomer of EE3M2H -.•- Z-EE3M2H --- 10Z:1E 3M2H Adaptation Recovery I I., I, "l / i- ,I I I .t"ll--oT"l 'l i!l'i-i-iii', ,,,!./-K,•I, I ' I ' Time (min) Figure 4. Mean magnitude estimates (with standard errors) as a percentage of the initial estimates for the individual isomers and a 10Z:lE mixture of 3M2H following adaptation to each isomer. 33.8% and 22.2% for the two sessions using E-EE3M2H as the adapting odorant). Estimates were 36.7% and 38.3% for the two sessions using Z-EE3M2H and continued for the duration of the adaptation period (estimates did not vary significantly throughout the adaptation period for any self-adapted odorant). Following removal of the adapting odorant, magnitude estimates were significantly greater than estimates during self-adaptation for both ethyl ester isomers. Each ester isomer displayed a pattern of recovery to baseline levels in one session, and incomplete recovery in the other session (i.e., estimates during recovery remained significantly lower than initial estimates Tables I and II). Cross-adaptation. The ethyl ester isomers were differentially successful in cross-adapting the 3M2H mixtures. Exposure to the E-isomer of EE3M2H significantly reduced the
372 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Table I Mean Magnitude Estimates as a Percentage of Initial Estimates and Associated F-Values for the Comparison of Mean Magnitude Estimates for the 10:1 E:Z 3M2H Mixture and the Individual Ethyl Ester Isomers During and Following Adaptation to Each of the Ethyl Ester Isomers Adaptation Recovery Odorant X (S.E.) F X (S.E.) F Exposure to (E)-EE3M2H 10:1 E:Z 3M2H 41.9% (8.45) 47.28*** 59.8% (8.99) 20.05*** (E)-EE3M2H 33.8% (9.08) 53.18'** 85.2% (9.68) 2.32 Exposure to (Z)-EE3M2H 10:1 E:Z 3M2H 72.1% (12.15) 5.27 66.9% (9.77) 11.46' (Z)-EE3M2H 36.7% (7.13) 78.82*** 76.8% (3.88) 35.76*** *p .01. **p .005. ***p .001. The values in the table represent the mean magnitude estimate for an odorant expressed as a percentage of the initial estimates. Each F-test compares this mean magnitude estimate with the initial magnitude estimates for that odorant. Degrees of freedom for all F-tests = (1,11). The significance level was set at p .01 because multiple F-tests were performed. Table II Mean Magnitude Estimates as a Percentage of Initial Estimates and Associated F-Values for the Comparison of Mean Magnitude Estimates for the 10:1 Z:E 3M2H Mixture and the Individual Ethyl Ester Isomers During and Following Adaptation to Each of the Ethyl Ester Isomers Adaptation Recovery Odorant X (S.E.) F X (S.E.) F Exposure to (E)-EE3M2H 10:1 Z:E 3M2H 58.4% (7.35) 32.02*** 71.0% (6.56) 19.53'** (E)-EE3M2H 22.2% (3.66) 452.16'** 75.5% (7.65) 10.24' Exposure to (Z)-EE3M2H 10:1 Z:E 3M2H 67.0% (11.43) 8.33* 80.2% (8.68) 5.20 (Z)-EE3M2H 38.3% (9.42) 42.90*** 92.1% (9.65) 0.68 *p .01. **p .005. ***p .001. The values in the table represent the mean magnitude estimate for an odorant expressed as a percentage of the initial estimates. Each F-test compares this mean magnitude estimate with the initial magnitude estimates for that odorant. Degrees of freedom for all F-tests = (1,11). The significance level was set at p .01 because multiple F-tests were performed. perception of both 3M2H mixtures via cross-adaptation. Thus, estimates for the 10:1 E:Z mixture were reduced to 41.9% of initial estimates following exposure to the E-isomer (Figure 3, Table I), a level comparable to that observed for the E-isomer self-adaptation. Estimates for the 10:1 Z:E mixture were also reduced following exposure to the E-isomer, albeit not as strongly (58.4% of initial estimates Figure 4, Table II). Estimates for each of the 3M2H mixtures showed partial recovery following removal of the adapting E-isomer, but remained significantly depressed relative to initial estimates
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)