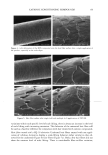
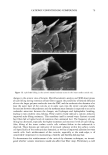
104 JOURNAL OF COSMETIC SCIENCE CLINICAL EFFICACY OF 2% POLYPHENONE (GREEN TEA EXTRACT) IN A HYDROPHILIC GEL ON AGED AND DAMAGED FACIAL SKIN Tanweer A. Syed •, Ph.D., Wendy Hiu-Wai Wong 2 and Seyed A. Ahmad 3 •Department of Dermatology, University of California San Francisco 2BWT Group, Inc., 1032 Irving Street, #917, San Francisco, CA 94122 SDepartment of Chemistry, University of California, Berkeley, CA Introduction: Polypenolic Ëactions and the chemistry of -EGCg (Epigallocatechin gallate) or green tea extract molecules exhibit a variety of antioxidants that can regulate cell division, proliferation, platelet aggregation, and detoxification (1,2). Obviously, such properties can significantly contribute to impart clinically beneficial response to stimulate the dermis to form new collagen and elastin (the support fibers that prevent the skin from sagging and wrinkling) and also help in slowing or reversing the aging process. Additional, clinical data has also shown that green tea extract can also inhibit the release of catecholamines and the formation of SNARE complex (3,4). Objective: To evaluate the clinical efficacy and beneficial effects of 2% (-EGCg) green tea extract in a hydrophilic gel (1% hydroxyethylcellulose solution) on aged and damaged facial skin. Methods: Preselected, female panelist (n=60), aged 25-60 years showing visible signs of damaged facial skin observed by uneven skin texture and large pores were sequentially randomized into two parallel groups (active and placebo). Each subject was allocated an identical pre-coded tube (50-g) with instructions on how to topically apply the trial gel two times a day for 4 weeks. Clinical efficacy and beneficial effects were assessed by dermatological evaluations, such as, irritation, changes in skin firmness and elasticity and instrumental measurement of skin hydration. Photographic and optical techniques were used both at the baseline and on a weekly basis. Efficacy data regarding anti-aging and damaged facial skin was evaluated by both in vitro and in vivo tests. Inhibition in the release of catecholamines was ascertained by monitoring the neurotransmitters Adrenaline and Noradrenaline. Chromaffin cells (prepared from bovine adrenal glands by collagenase digestion and separated from erythrocytes) were incubated with adrenalin/noradrenaline and the trial gel. Both the release of catecholamines and the total cell count were determined by liquid scintillation. Evaluation of SNARE complex inhibition was also conducted on chromaffin cells. Skin topography analysis (in vivo) was performed by obtaining silicon imprints from around the eyes and the damaged facial skin of the subjects. Such imprints were obtained at 0 (baseline), 15 and after 30 days of two times a day regimen. Analysis of the imprints were performed by confocal laser scanning microscopy. The tolerability of the study gel was assessed at the scheduled visit by direct questioning of the subjects about study gel related subjective and objective adverse symptoms. For safety and toxicological evaluation, cytotoxicity tests on human epidermal keratinocytes and human dermal fibroblasts were also performed before the initiation of the trial. An informed consent was obtained from all the subjects. Study participants were requested not to use any emollient or topical application during the study period. The X • and Fisher's exact tests, with two-tailed figures were used for significant test results.
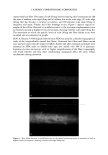
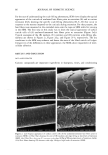
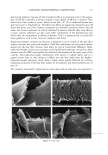
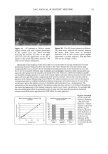
2002 ANNUAL SCIENTIFIC MEETING 105 Results: All the sixty subjects were available for efficacy analysis. The regimen received full subject's compliance with no drop outs. By the end of the treatment marked beneficial improvement in facial skin texture and appearance was observed in 45% subjects. Code disclosure revealed that blend of 2% -EGCg in gel significantly improved facial and damaged skin of 23/30 (76.7%) while placebo benefited 4/30 (13.3%) subjects respectively (2% EG-Cg gel versus placebo, p0.0001). Data from liquid scintillation counting for catecholamine release showed that 2% green tea extract gel significantly inhibited Adrenaline and Noradrenaline neurotransmitters at 85% and 52% nM concentrations against control sample. In case of SNARE complex analyzed immunocomplexes proved to prevent vesicle docking by inhibiting the formation of the essential ternary SNARE complex. Skin topography analysis showed that the severity ofwrinldes around the eyes and the aging of the facial skin decreased up to 45% after 15 days of treatment and up to 88% after 30 days of treatment. In vitro cytotoxicity on human epidermal keratinocytes at concentrations between 10pg/mL and 1 mg/mL with a keratinocyte density of 15,000 cell/cm showed no signs of cytotoxicity. Cytotoxicity tests on human derreal fibroblasts at concentrations between 10pg/mL and 1 mg/MI with a cell density of 21,000 cell/cm 2 showed no cytotoxicity. Subjects complained of no side effects. Conclusion: The study demonstrates that 2% polyphenone (-EGCg, green tea extract) in a hydrophilic gel is safe and significantly more beneficial than placebo to impart superior clinical efficacy on aged and damaged facial skin. Reference•: Balentine DA, Wiseman SA, Bouwens LCM. Crit Rev Food Sci Nutr 37:693-704, The chemistry of tea flavonoids (1997). Beechef GK, Warden BA, Metken H. P.S.EB.M 220:267-270, Analysis of Tea Polyphenols (1999). Gutierrez LM, Viniegra S, Rueda $, Ferr-Montiel AV. $ Biol Chem 272:2634-9, A peptide that mimics the C-terminal sequence of SNAP-25 inhibits secretory Vescle docking in chromaffin cells (1997) Syed TA, Wong Hiu-Wai W, Ahmad SA, Fatima S. TMIH (in press). Clinical evaluation of 2% Polyphenone (green tea extract) stabilized in a hydrophilic gel to improve facial wrinkles (2002).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)