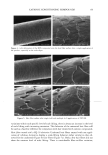
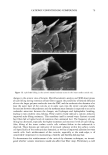
EPIDERMAL PENETRATION OF SDS MICELLES 31 stated that the observed dose dependence of surfactant-induced skin irritation beyond the CMC cannot be explained solely by the contribution of the monomeric surfactant, and speculated that the dose-dependence may result from the penetration of submicellar aggregates into the skin (32). However, in this paper, we present evidence that the mice//ar surfactant contributes to surfactant penetration into the skin, and investigate the factors that control the penetration of both miceliar and monomeric surfactant into the epidermis. With this in mind, we have measured the amount of epidermis, consisting of the SC and the viable epidermis, aqueous SDS solutions of increasing SDS concentration. centration of SDS in the epidermis is directly related to micelies in the contacting solution. We have also found SDS that penetrates into the after five hours of exposure to We have found that the con- the concentration of free SDS that, in the presence of poly- (ethylene oxide), the SDS that complexes with PEO in the form of PEO-bound SDS micelies cannot penetrate into the epidermis, while the free, or unbound, SDS micelies can. Using dynamic light scattering (DLS), we will show that a plausible explanation of our findings is that the free SDS micelies can penetrate into aqueous pores present in the SC, while the PEO-bound SDS micelies are sterically hindered from penetrating into the SC. EXPERIMENTAL MATERIALS Sodium dodecyl sulfate (SDS) and poly(ethylene oxide) (PEO) (molecular weight of 8000 g/mol) were purchased from Sigma Chemical Company (St. Louis, MO) and used as received. Water was produced using a Millipore Academic water filter. •4C-radiolabeled SDS was purchased from American Radiolabeled Chemicals (St. Louis, MO) and used as received. Phosphate-buffered saline (PBS) was prepared using PBS tablets from Sigma and Millipore-filtered water. PREPARATION OF SKIN SAMPLES Female Yorkshire pigs (40-45 kg) were purchased from local farms, and the skin (back) was harvested within one hour after sacrificing the animal. The subcutaneous fat was trimmed off using a razor blade, and the full-thickness pig skin was cut into small pieces and stored in a -80øC freezer for up to two months. The surfactant penetration experi- ments were performed using full-thickness pig skin. EXPERIMENTAL PROTOCOL The pig skin was mounted in a vertical Franz diffusion cell (Permegear Inc., Riegelsville, PA), with the SC side facing the donor compartment. Phosphate-buffered saline (PBS phosphate concentration of 0.01 M NaCI concentration of 0.137 M Sigma Chemical Company, St. Louis, MO) was added to the donor and the receiver compartments of the diffusion cell, and the skin was allowed to hydrate for one hour. The PBS in the donor compartment was removed, and 1.5 ml of surfactant solution was added to the donor
32 JOURNAL OF COSMETIC SCIENCE compartment. The solution in the donor compartment, referred to hereafter as the contacting solution, contained mixtures of SDS and PEO, each with about 0.5 pCi/ml of 14C-SDS. We verified that the concentration of radiolabeled SDS in the contacting solution did not change appreciably during the five-hour exposure to the skin. Diffusion into the skin took place for five hours, and subsequently the contacting solution was removed and the donor compartment was rinsed four times with 2 ml of PBS. A five-hour exposure was chosen because this was a sufficiently long time to allow significant SDS penetration into the skin, but a short enough time to prevent the saturation of the skin with SDS. The temperature of the diffusion cell was ambient, that is, 20 + IøC. The skin was then heat-stripped by placing it in a bath of water at 60øC for two minutes, and then peeling off the epidermis (SC and viable epidermis) that had been exposed to the contacting solution from the dermis. The exposed epidermis was then dried for two days in a fume hood and weighed. The dried epidermis was dissolved overnight in 1.5 ml of Soluene-350 (Packard, Meriden, CT). After the epidermis dis- solved, 10 ml of Hionic Fluor scintillation cocktail (Packard) was added to the Soluene- 350, and the concentration of radiolabeled SDS was determined using a Packard Tri- Carb 4350 scintillation counter (Packard). Knowing the concentration of SDS in the contacting solution, Csos, the radioactivity of the contacting solution, Crad, donor, the dry weight of the epidermis, m, and the radioactivity of the epidermis, Cr•,skin , it was possible to determine the concentration of SDS in the dried epidermis, CSOS, skin , using the following equation: C r•d, •kin ' CSDS (1) C SDS, skin z Grad, donor ' m DYNAMIC LIGHT SCATTERING The SDS and SDS+PEO solutions were prepared in Millipore-filtered water with 0.1 M NaCI. After mixing, the solutions were filtered through a 0.02 pm Anotop 10 syringe filter (Whatman International, Maidstone, England) directly into a cylindrical- scattering cell to remove any dust from the solution, and then sealed until use. Dynamic light scattering (DLS) (33) was performed at 25øC and a 90 ø scattering angle on a Brookhaven BI-200SM system (Brookhaven, Holtsville, NY) using a 2017 Stabilite argon-ion laser (Spectra Physics) at 488 nm. The autocorrelation function was analyzed using the CONTIN program provided by the BIC dynamic light scattering software -- (Brookhaven, Holtsville, NY), which determines the effective hydrodynamic radius, using the Stokes-Einstein relation (34): kBT -- Rh - 6q. rxl• (2) where kB is the Boltzmann constant, T is the absolute temperature, x I is the viscosity of -- the salt solution, and D is the mean diffusion coefficient of the scattering species. In order to measure the size of the micelies, including free SDS micelles and PEO-bound SDS micelies, and eliminate the effects of interparticle interactions, the effective hydro- dynamic radii were determined at several different SDS concentrations, and the average effective hydrodynamic radii were extrapolated to a zero micelie concentration (33,35-
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)