20 JOURNAL OF COSMETIC SCIENCE (27) (28) (29) (5) M. Tanaka, Recent development of skincare products in Japan from the bio-science standpoint, FragranceJ., 14, 9-15 (in Japanese) (1994). (6) R. S. Summers, B. Summers, P. Chandar, C. Feinberg, R. Gursky, and A. V. Rawlings, The effect of lipids, with and without humectant, on skin xerosis, J. Soc Cosmet. Chem, 47, 27-39 (1996). (7) S. Matsumura, H. C. Cheng, M. Minami, S. Yoshikawa, and T. Karltoni, Synthesis and water uptake and holding capacity of chitin and chitosan derivatives, Oil Chem., 38, 492-500 (1989). (8) E. Vandamme, Bacteria in front mirror: Biocosmetics vis microbial synthesis, Agro Food Industry Hi. Tech., 3-8 (July/August 1996). (9) S. B. Ross-Murphy, "Properties and Uses of Cellulose Solution," in Ce//u/ose Chemistry and Its Appli- cations, T. P. Nevell and S. H. Zeronian, Eds. (Ellis Horwood, West Sussex, UK, 1985), pp. 202-222. (10) T. L. Davidson, Handbook of Water-Soluble Gums and Resins (McGraw Hill, New York, 1980). (11) G.L. Brode, "Polysaccharides: 'Naturals' for Cosmetics and Pharmaceuticals," in Cosmetic and Phar- maceutical Applications of Po/ymers, C. G. Gebelein, T. C. Cheng, and V. C. Yang, Eds. (Plenum, New York, 1991), pp. 105-115. (12) R. H. Chen and R. S. Heh, Sun protection and skin hydration effects and physico-chemical properties of sun screen lotion containing water-soluble chitosan, J. App/. CosmetoL, 17, 56-71 (1999). (13) R. H. Chen and R. S. Heh, Film formation time, skin hydration effects, and physicochemical prop- erties of moisture masks containing different water-soluble chitosans,J. Cosmet. Sci., 51, 1-13 (2000). (14) R. H. Chen and R. S. Heh, Skin hydration effects, physico-chemical properties and vitamin E release ratio of vital moisture creams containing water-soluble chitosan, Int. J. Cosmet. Sci., 22, 349-360 (2000). (15) N. Mekideche and N. Briand, Out of the blue soap, Perfum. Cosmet., 67, 73-78 (1994). (16) H. Iida, K. Nakamura, and T. Tokunaga, Dimethyl sulfide and dimethyl-13-propiothetin in sea algae, Bu//. Jpn. Sot Sci. Fish., 51, 1145-1150 (1985). (17) T. Kajiwara, A. Hatanaka, T. Kawai, M. Iahihara, and T. Tuneya, Long chain aldehydes in the green marine algae Ulvaceae, Nippon Suisan Gakkaishi, 53, 1901 (1987). (18) H. Sakagami, J. Iseda, T. Fujimori, Y. Hara, and M. Chihara, Volatile constituents in marine algae, Nippon Suisan Gakkaishi, 57, 527-533 (1991). (19) K. Fujimoro and T. Kaneda, Screening test for antioxygenic compounds from marine algae and fractionation from Eisenia bicyclis and Undaria pinnatifida, Bull. Jpn. Soc. Sci. Fish, 46, 1125-1130 (1980). (20) J. Tsai and S. Lin, Emulsifying properties of Monostroma nitidium mucilage-protein conjugates,J. Chin. Agric. Chem. Soc., 33, 104-113 (1995). (21) J. Tsai and T. Chiou, Effect of enzymatic hydrolysis on the extraction yield and emulsifying properties of Monostroma nitidium hydrolysate. J. Chin. Agric. Chem. Soc., 34, 241-247 (1996). (22) R. H. Chen and W. Y. Chen, Rheological properties of the water-soluble mucilage of a green laver, Monostroma nitidium, J. Appl. Phycol., 13, 481•488 (2001). (23) AOAC (Association of Official Analytical Chemists), OfjS'cial Methods of Analysis, 14th ed. S. Williams, Ed. (AOAC, Washington DC., 1984). (24) F. Draize, G. Woodard, and H. O. Calvery, Appraisal of the safety of chemicals in food, drugs and cosmetics, Pharmacology, 93, 377-392 (1948). (25) J. L. Leveque and J. De Rigal, Impedance methods for studying skin moisturisation, J. Soc. Cosmet. Chem., 34, 419•428 (1983). (26) W. Gehring, J. Wenz, and M. Gloor, Influence of topically applied creamide/phospholipid mixture on the barrier function of intact skin, atopic skin and experimentally induced barrier damage, Int. J. Cosmet. Sd., 19, 143-156 (1997). Y. Yazan, K. Arslan, and M. Seiller, Formulation and evaluation of a multiple emulsion containing glycolic acid, Drug Cosmet. Ind., 160, 30-37, 101-103 (1997). C. van den Hoek, D. G. Mann, and H. M. Jahns, "Pigments and Chloroplasts," in Algae (Cambridge University Press, Cambridge, UK), pp. 337-338. R. A. Muzzarelli, M. Weckx, O. Filippini, and C. Lough, Characteristic properties of N-carboxybutyl chitosan, Carbohydr. Polym. 11, 307-320 (1989).
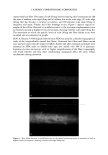
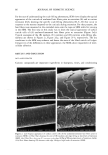
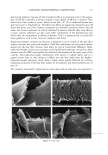
j. Cosmet. sci., 54, 21-27 (January/February 2003) Analysis of DNA in hair fibers DANIELLA M. HEYWOOD, RICHARD SKINNER, and PAUL A. CORNWELL, Hair Group, Unilever Research, Port Sunlight, Quarry Road East, Bebington, Wirral, CH63 3JW, United Kingdom. Accepted for publication September 6, 2002. Synopsis The extraction and identification of deoxyribonucleic acid (DNA) from human hair shafts is described, along with the effects of hair treatments on levels of DNA and suggestions of DNA location within the shaft. DNA was present at low levels in the hair shaft, and was identified using polymerase chain reaction amplification of the human leukocyte anrigen (HLA)-DQA1 locus. The use of cleanup columns aided the success of PCR amplification. DNA appears to reside in the cuticle portion of the hair shaft. Levels of DNA were found to be higher at the root-end compared to the tip-end of hair and were also found to be lower after permanent colorant treatment. DNA was found to be lost with surfactant washing, with increased loss occurring with prolonged or an increasing number of washes. These results suggest that small amounts of residual DNA remain after differentiation and add to our knowledge of the constituents of hair. INTRODUCTION The analysis of deoxyribonucleic acid (DNA) from intact roots of single hair fibers is now routinely used in the area of forensic science. While the detection of mitochondrial DNA from hair shafts is quite successful (1), the detection of genomic DNA from hair shafts without the presence of an intact root is far less reliable, but has been carried out by a few research groups (2-5). Contrary to previous thoughts, results demonstrating the presence of high-molecular-mass genomic DNA in hair shafts (5) suggest that not all genomic DNA degenerates during the keratinization process. The level of DNA in hair shafts has been shown to be residual compared to the average 0.5 l•g found in intact hair follicles (6). These low levels, combined with the fact that water-soluble melanins, responsible for the pigment of hair, inhibit DNA polymerase enzymes, make DNA analysis from hair shafts difficult (7). These water-soluble melanins are produced spontaneously within the hair fiber, but levels are provoked by hydrogen peroxide, a component of permanent colorants and bleaches (7). Address all correspondence to D. M. Heywood. 21
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)