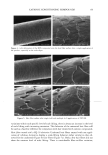
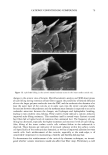
EPIDERMAL PENETRATION OF SDS MICELLES 33 38). We added 100 mM of NaCl to screen the intermicellar electrostatic interactions in the DLS measurements (33,36-38). In the presence of PEO, the concentration of PEO was adjusted such that only PEO- bound SDS micelies were predicted to exist (39). This was done to minimize the scattering from either the free SDS micelies or the free PEO molecules, both of which have R3 values that are similar to that of the PEO-bound SDS micelies. The concen- trations of SDS and PEO utilized in the DLS studies are listed in Table I. DETERMINATION OF THE AQUEOUS PORE RADIUS IN THE STRATUM CORNEUM Although we did not measure the aqueous pore radii ourselves, we believe that a brief discussion of the measurement method is in order. Using hindered-transport theories (40), several authors have measured the average aqueous pore radius in the SC (12,41- 44). In this method, the transdermal permeabilities of two types of polar molecules, or the transdermal permeability of a polar molecule and the transdermal conductivity of an ion, are measured. The permeants are either both radiolabeled molecules (12,43) or a radiolabeled molecule and an ion (42,44), which have different hydrodynamic radii. The effect of the skin pore radius on the diffusion of the permeant depends on the ratio of the permeant hydrodynamic radius to the pore radius, )•, with an increase in the hindrance to the diffusion of the permeant across the skin as )• approaches 1. Accordingly, by comparing the hindrance to diffusion across the skin of the two molecules, one can determine the effective aqueous pore radius of the skin. RESULTS AND DISCUSSION DOSE-DEPENDENT PENETRATION OF SDS INTO THE EPIDERMIS We first investigated whether increasing the total SDS concentration in the contacting solution above the CMC led to an increase in the concentration of SDS measured in the epidermis. An increase in the SDS concentration in the epidermis could explain much of the skin irritation data reported in the literature, since a larger "dose" of the SDS irritant in the skin would lead to greater skin irritation. Figure 1 shows that as the concentration of SDS in the contacting solution increased from 8.7 mM (the CMC of SDS) to 200 mM (about 20 times the CMC of SDS), the concentration of SDS measured in the epidermis increased in a linear manner, similar to the increased penetration reported by others (28,29). This finding clearly contradicts the monomer penetration model, according to which the amount of SDS penetrating into the epidermis should Table I Concentrations of SDS and PEO Used in the Dynamic Light-Scattering Experiments to Minimize the Concentrations of Free SDS Micelles and Free PEO Molecules in the Scattering Solution Concentration of SDS (mM) Concentration of PEO (wt%) 10 0.14 20 0.29 30 0.45 4O O.6O
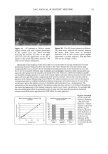
34 JOURNAL OF COSMETIC SCIENCE 12 :i• 6 o 4 o 2 CMC I 0 50 100 150 200 SDS Concentration in the Contacting Solution (mM) Figure 1. Effect of increasing the SDS concentration in the contacting solution on the amount of SDS that penetrates into the epidermis. The vertical dashed line indicates the CMC of SDS (8.7 mM), and the solid line through the data points is shown to guide the eye. The error bars reflect a 95% confidence interval based on six samples at each SDS concentration. remain constant as the SDS concentration increases beyond the CMC. Instead, Figure 1 vividly demonstrates that SDS present in miceliar form must be contributing to SDS penetration into the epidermis, since the SDS monomer concentration is constant over the experimental range examined, while the concentration of SDS micelies is increasing. The concentration of SDS in the epidermis measured here (2-9 wt%) compares well with the concentrations reported by others (4.3 wt%) (45). This large partitioning of SDS into the skin has been viewed as reflecting the binding of SDS molecules to the keratin protein in the corneocytes found in the SC (28,29,45). A mass balance of the amount of SDS found in the epidermis relative to the initial amount of SDS indicates that less than 1% of the SDS in the initial contacting solution penetrates into the skin for all the contacting solution used in Figure 1. Accordingly, one can assume that the contacting solution provides an infinite reservoir of SDS for penetration into the skin. Although the results in Figure 1 contradict the monomer penetration model, they do not contradict many observations reported in the literature where a surfactant dose- dependent skin irritation response was observed (1-3,7,10,13,15). Moreover, if the surfactant-induced damage to the skin is related to the actual amount of SDS in the skin, then the increased concentration of SDS in the epidermis shown in Figure 1 can be related to the increased skin irritation induced by SDS, observed by many researchers, as the SDS concentration in the contacting solution is increased (1-3,7,10,13,15). EFFECT OF PEO ON THE PENETRATION OF MICELLAR SDS INTO THE EPIDERMIS Having demonstrated that the amount of SDS penetrating into the epidermis above the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)