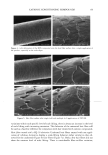
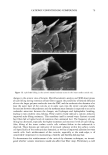
10 JOURNAL OF COSMETIC SCIENCE tants used in moisture products, occlusive humectants form a hydrophobic film to retard the evaporation of the water from the skin surface. Common occlusive humectants are petrolatum, fatty acids, and cholesterols (6). The second type of humectant is an ab- sorbing humectant. These compounds have strong water-absorbing properties and will prevent dryness and cracking of the skin in low-humidity environments. Common absorbing humectants are glycerin, propylene glycols, and high-molecular-weight hu- mectants such as hyaluronic acid and chitinous materials (7,8). Ross-Murphy (9) reported that cellulose was applied to shampoo and hair conditioner as a suspension and viscosity enhancer. Davidson (10) reported the industrial use of car- rageenan as a binder in toothpaste and as a bodying agent to provide slip and emollience in lotions and creams. Brode (11) reported that polysaccharides have been applied in many cosmetic products such as skin moisturizers or conditioners, soaps, sunscreens, cleansing creams or body lotions, gels or lotions, toothpastes, foam builders, mousses, shampoos, and nail lacquers. Chen and Heh (12-14) reported that water-soluble chitosan improved the efficacy of sunscreen lotions, moisture masks, and vital moisture creams. Mekidecke and Briand (15) reported that seaweed extracts provide an anti-aging effect and a fat-removing effect, as well as maintaining elasticity of the skin, preventing skin aging, and providing a sun-screening effect. Therefore, seaweed extracts are excellent cosmetic ingredients. Since it is the precursor of dimethylsulfide, one of the desirable and characteristic seaweed aromas, dimethyl-[B-propiothein is abundant in green algae and can be effec- tively extracted by the use of a hydrophilic solvent from dried algae (2,16-18). Chlo- roform-soluble and aqueous fraction show anti-oxidation capacity (19). Water-soluble mucilage of Monostroma nitidium shows good emulsifying properties (20,21) and thick- ening properties (22). The mucilage of Monostroma nitidium may be applied as a natural, renewable cosmetic ingredient. Therefore, the effects of using the aqueous extracts of Monostroma nitidium to replace half of the thickening agents and humectants in a mois- ture mask formula on the rheological properties, color, storage stability, water-holding capacity, and film formation times of moisture masks were explored. MATERIALS AND METHODS PREPARATION OF MONOSTROMA NITIDIUA4 AQUEOUS EXTRACT We washed the dried alga to remove impure materials such as sand. We added 50 volumes of distilled water, homogenized the mixture briefly for 3 min with a blender (Osterizer), then homogenized it again at 9000 rpm (Polytron PT 300, Kinematic AG, Switzerland) for 7 more min. We heated the homogenized solution, stirring in an oil bath at 100øC for 4 h. We centrifuged the heated homogenates at 3500 x g for 30 min (Hitachi SCR20BA, Hitachi Co., Tokyo). We discarded the residues and lyophilized the supernatant with a freezer-dryer (UNITOP 800L Virtis Company Inc., USA) to obtain the products (4,20). The approximate compositions were determined with the AOAC method (23). PREPARATION OF MOISTURE MASK The formula for the moisture mask is shown in Table I. Monostroma nitidium water- soluble mucilage (0.5% to 2.0% w/v) was used to replace the thickening agent (2.0%
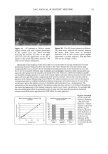
WATER-SOLUBLE MUCILAGE IN A MOISTURE MASK 11 Table I Formula for Moisture Mask Ingredients Chemicals Percentage (%) Film-forming agent Polyvinyl alcohol 15.0 Thickening agent Methyl cellulose 2.0 Humectant 1-3 butadiene 5.0 Alcohol Ethyl alcohol 12.0 Preservative Methylparaben 0.4 Buffer Sodium citrate q.s. for pH 6.2-6.4 Surfactant Polyoxyethylene 20 oleyl ether 0.5 Water q.s. 100 methyl cellulose) and humectant (5% 1,3 butadiene). The procedures were: The buffer and humectant were added to the water and heated at 70-80øC, and then the thickening agent and film-forming agent were added and mixed well to form the aqueous phase at 70-80øC. The preservative and surfactant were added to ethyl alcohol to obtain the oil phase at 70-80øC. The oil phase was poured into the aqueous phase and homogenized to obtain the product (13). CHARACTERISTICS OF MOISTURE MASKS CONTAINING AQUEOUS EXTRACT Viscosity of moisture mask. A 5-ml aliquot of moisture mask was placed onto a plate and plate cell (PQ 45), which was maintained at 25øC. Viscosity measurements were per- formed at shear rates of 0-100 s i with a Haake viscometer (model CV 20, Haake Mess-Technik Gmbh., Germany). Saj•ty test. The safety of the product was tested in terms of the sensitivity of shaved rabbit (NZW) skin. A 0.1-g aliquot of prepared moisture mask was evenly applied to ca. a 9-cm 2 patch of shaved rabbit skin. Rabbits were kept in an air-conditioned room (25 ø + 0.5øC and 50-60% RH). The sensitivity of the rabbit skin was evaluated by the Draize scoring method (24). Water-holdi,g capacity test. The water-holding capacity of the skin was tested by the corneometer method (25-27). The corneometer measures changes of electrical capaci- tance that are related to the moisture content of the skin before and after applying the moisture mask. A 0.2-g aliquot of moisture mask was evenly applied to ca. a 25-cm 2 patch on the volar forearms of seven healthy volunteers, who were fully informed of the nature of the study and the procedure involved, in a test room of RH 60% + 5% and 23 ø + 0.5øC. Changes of electrical capacitance were recorded over time with a skin analyzer SHP88 (sebumeter + corneometer + pH-meter, Courage and Khazaka, Ger- many) randomly at eight points. Every sample measurement was repeated three times. The capacitance increase ratio was expressed as: Electrical capacitance after moisture mask applied Capacitance increase ratio = Electrical capacitance of skin without moisture mask The original electrical capacitances were between 13.4 and 14.2 corneometer units. Film formation time. A 0.2-g aliquot of moisture mask was applied by an experienced person to ca. a 2 5-cm 2 patch on the volar forearms of seven healthy volunteers (who were
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)