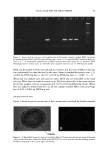
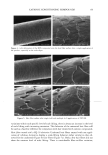
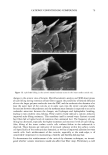
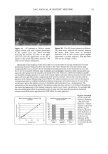
EPIDERMAL PENETRATION OF SDS MICELLES 35 CMC is related to the total SDS concentration (miceliar and monomeric) in the con- tacting solution, instead of only to the SDS monomer concentration, we examined the effect of adding PEO to the contacting solution. PEO is known to form micelie-like complexes with SDS, with the PEO forming a corona around the SDS micelies (35,38,46). The critical aggregation concentration (CAC) is the surfactant concentration at which polymer-bound micelies first form, in our case PEO-bound SDS micelies, and is lower than the CMC of the surfactant in the absence of PEO (39). When PEO is mixed with a solution of SDS, if there is an excess of PEO, the SDS monomer concentration is approximately equal to the CAC. Consequently, we anticipated that the contribution of the SDS monomers to skin penetration would be diminished in the SDS-PEO solutions because the SDS monomer concentration would be lowered. However, it was unclear how the SDS in the PEO-bound SDS micelies would behave when exposed to the skin, namely, whether this SDS could contribute to SDS penetration into the epidermis. Initially, we prepared solutions containing either 50 mM or 100 mM SDS as well as 2.0 wt% PEO. According to a molecular-thermodynamic theory developed recently (39), there is an excess of PEO at the two SDS concentrations examined. As a result, any micelies that form are bound to the PEO, and the SDS monomer concentration should be approximately equal to the CAC. Moreover, for PEO having a molecular weight of 8000 g/mol, we would expect, according to the theory (39), that there would be a maximum of 1 SDS micelie per PEO molecule. Figure 2 shows how the concentration of SDS measured in the epidermis was affected by the presence of PEO. Two aspects of Figure 2 should be noted: i. The addition of PEO at a given SDS concentration (50 mM or 100 mM) in the contacting solution leads to a significant reduction in the concentration of SDS measured in the epidermis. ii. The difference between the concentrations of SDS in the epidermis at 50 mM and • 6 •-•'4 0.•_ 0 3 I: 2 I: 1 0 0 ii I SDS SDS+PEO 50 mM 50 mM SDS , SDS+PEO 100 mM 100 mM Figure 2. Effect of adding 2 wt% PEO to a solution of SDS (50 mM and 100 mM) on the amount of SDS that penetrates into the epidermis. The empty bars indicate SDS penetration in the absence of PEO, and the solid bars indicate SDS penetration in the presence of 2 wt% PEO. The error bars reflect a 95% confidence interval based on six samples at each condition.
36 JOURNAL OF COSMETIC SCIENCE 100 mM SDS concentrations in the contacting solution is not statistically significant in the presence of PEO, while it is statistically significant in the absence of PEO. According to the monomer penetration model, the reduction in the SDS monomer concentration upon the addition of PEO should lead to a reduction in the amount of SDS penetrating into the epidermis, thus potentially explaining (i). However, the more interesting aspect of Figure 2, (ii), is that SDS bound to PEO contributes very little, if at all, to the amount of SDS measured in the epidermis as the SDS concentration increases from 50 mM to 100 mM, while the SDS in free micelies does. Instead, it is apparent that increasing the SDS concentration in the presence of PEO has no effect on the amount of SDS that penetrates into the epidermis. Therefore, SDS penetration into the skin in the presence of PEO does not increase with total SDS concentration, and follows the prediction of the monomer penetration model. In other words, the addition of PEO modifies the penetration behavior of the miceliar SDS, in effect reducing the system to one that obeys the monomer penetration model. To further study the relationship between the various forms of SDS present in the contacting solution (SDS monomers, free SDS micelies, and PEO-bound SDS micelies) and the SDS concentration measured in the epidermis, the skin was exposed to solutions of 100 mM SDS and different PEO concentrations. The PEO concentration in the contacting solution was varied such that there would be different proportions of free SDS micelies and SDS micelies bound to PEO, thereby controlling the concentration of SDS in free micelies while maintaining a constant total SDS concentration, including SDS monomers, free SDS micelies, and PEO-bound SDS micelies. The effect of increasing the total PEO concentration on the distribution of SDS between free SDS micelies and PEO-bound SDS micelies is shown schematically in Figure 3, with Figure 4 showing how the predicted concentrations of free SDS micelies and PEO-bound SDS micelies vary with increasing PEO concentration at 100 mM SDS (39). Figures 3 and 4 reveal that as the concentration of PEO increases, the concentration of free SDS micelies decreases as more SDS forms PEO-bound SDS micelies. However, as long as free SDS micelies are able to form, the SDS monomer concentration is predicted to remain constant with increasing PEO concentration. This is because once PEO is saturated with SDS micelies, the SDS monomer concentration should increase until the CMC is reached, beyond which free SDS micelies will form (39). However, above a concentration of 1.75 wt% PEO, no free SDS micelies are present, and the SDS monomer concentration is equal to the CAC of SDS in the presence of PEO, -5 mM (39). This was, in fact, the condition under which the measurements reported in Figure 2 were actually carried out. Figure 5 clearly shows that the concentration of SDS in the epidermis decreases as the concentration of PEO in the contacting solution increases, even though the total con- centration of SDS is fixed at 100 mM. Figure 5 also compares this experimentally observed decrease with the ]9redicted decrease in the concentration of SDS in free micelies with increasing PEO concentration, also shown in Figure 4 (39). The observed corre- lation between the concentration of free SDS micelies and the concentration of SDS measured in the epidermis is apparent, clearly demonstrating that SDS in the form of free micelies contributes to the penetration of SDS into the epidermis, while SDS in the form of PEO-bound SDS micelies does not. Although PEO prevents the SDS bound to it from penetrating into the epidermis, the SDS monomer can still penetrate into the epidermis, leading to the plateau observed at PEO concentrations above 1.75 wt%.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)