EPIDERMAL PENETRATION OF SDS MICELLES 41 Therefore, if micelie kinetics controlled the penetration of SDS into the epidermis in the presence of PEO, the penetration of SDS into the epidermis should increase as more SDS is added. Accordingly, one should observe an increase in the SDS concentration in the epidermis in Figure 2 as one goes from 50 mM SDS to 100 mM SDS in the presence of PEO. Instead, PEO prevents the PEO-bound SDS micelies from penetrating into the skin, as determined by the regression analysis presented above, thus making penetration model (i) inconsistent with the experimental observations. Penetration model (ii) assumes that the micelies are not stable when they approach the skin, and instead break apart when they impinge on the skin. In order for this mecha- nism to operate, the PEO-bound SDS micelies must somehow be prevented from con- tributing to this mechanism, while the free SDS micelles must be able to do so, evenwhen the two micelle types are presen simultanously in the conactin solution In refuting mechanism (i), we argued that the kinetics of SDS micelie dissolution are accelerated in the presence of PEO (51,52). This decreased micelle stability demonstrates that PEO-bound SDS micelies are less stable than free SDS micelies, and hence should be more likely to disintegrate as they impinge on the skin surface, which is contrary to the observed lack of contribution of the PEO-bound SDS micelies to SDS skin penetration. Accordingly, penetration model (ii) does not appear to adequately explain the observed dose- dependent penetration of SDS into the epidermis. Penetration model (iii) assumes that, contrary to the monomer penetration model, SDS micelies are actually able to penetrate into the SC. Although it is not expected that a SDS micelie would pass through the SC without breaking up due to its self-assembling nature, if the SDS micelies could penetrate even partially into the SC, then the concen- tration of SDS in the epidermis could be increased dramatically. If the free SDS micelles could penetrate into the SC while the PEO-bound SDS micelles could not, then the concentration of SDS in the epidermis would be related to the concentration of SDS in the free micelies and not to the concentration of SDS bound to PEO, as we observed experimentally in Figures 1, 2, and 5. Therefore, our hypothesis that some micelies can penetrate into the SC, while others cannot, appears to be consistent with our experi- mental observations. The concept proposed in this paper that micelies can penetrate into the skin differs from the earlier speculation that the submicellar aggregates are responsible for the observed dose dependence (32). In the case of SDS, the concentration of submicellar aggregates (aggregates composed of less than about 30 SDS molecules that do not constitute a complete micelie) does not increase significantly with total SDS concentration beyond the CMC (32). However, according to the results of our regression analysis presented above, the contribution of the non-monomeric fraction of SDS to SDS penetration into the skin (represented by b in Eq. 3) is between a third and a quarter of the monomeric contribution (represented by a in Eq. 3). The low concentration of submicellar aggre- gates, predicted by kinetic theories of micellization (48), is not sufficient to explain the large contribution of the free SDS micelies to SDS penetration into the skin. Indeed, the submicellar aggregates represent a very small fraction of the total non-monomeric SDS population, with the free SDS micelies accounting for the large majority of this popu- lation. If only the submicellar aggregates were responsible for the dose-dependent SDS penetration into the skin, then the rate of submicellar SDS penetration into the skin would have to exceed the SDS monomeric rate by several orders of magnitude to account for the observed SDS dose dependence. Finally, one would also need to explain why the
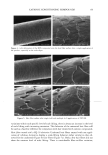
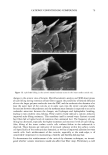
42 JOURNAL OF COSMETIC SCIENCE SDS submicellar aggregates contribute to SDS skin penetration only in the absence of PEO. Due to the hydrophilic nature of micelies, micelles would be expected to enter the SC through aqueous pathways (pores) rather than through non-polar pathways (53). Aque- ous pathways in the SC are believed to be located in the intercellular region, in particular in the lacunae and other aqueous regions surrounded by polar lipids (53-56). The characteristic radius of the aqueous pores in the skin has been determined using hin- dered-transport theories (12,42-44). Typical values for the aqueous pore radii of human skin range from 10 to 25 fit (12,43,44). For the Yorkshire pig skin used in this paper, the characteristic aqueous pore radius was determined to be about 28 fit (42). Peck eta/. (12) observed that the average pore size of the SC did not change significantly when the epidermis was exposed to SDS solutions for 18 hours. Instead, they concluded that the increased permeability of the skin resulted from an increase in the effective porosity/ tortuosity of the SC. This observation is relevant to our findings because it indicates that exposing the epidermis to an SDS solution will not change the aqueous pore sizes available for penetration into the skin. Nevertheless, we believe that additional research should be conducted to better understand the effect of surfactant penetration into the skin on the aqueous pathways of the SC. In Figure 6 we determined that the average radius of the free SDS micelies is 20 fit. These micelies are small enough that they could conceivably penetrate into the aqueous pores of the skin. However, according to our light-scattering studies, the PEO-bound SDS micelies have an average radius of 25 fit, and would be sterically hindered from passing into most aqueous pores in the skin since they are of about the same size as these pores. Consequently, according to our hypothesis, the ability of the micelies to affect the amount of surfactant penetrating into the skin is determined by the size of the micelie relative to the aqueous pore size. In this respect, it should also be recognized that both the micelies and the aqueous pores exhibit a range of sizes (31,57). It is expected that the micelies will only be able to penetrate into the skin when there is an overlap in these size ranges. Accordingly, although some aqueous pores may be large enough to allow the penetration of PEO-bound SDS micelies, such events would be relatively rare. In general, one should keep in mind that the aqueous pores in the skin are negatively charged (12,41). As a result, in addition to the micelie steric hindrance considerations put forward in this paper, there may be cases where electrostatic interactions between charged micelies and the charged aqueous pores need to be considered to determine the ability of charged micelies to penetrate into the aqueous pores. However, such electro- static effects are not expected to play a role in the studies reported in this paper because the free SDS micelies and the PEO-bound SDS micelies should have the same net negative charge, since PEO is a neutral molecule and its complexation with SDS does not screen the negatively charged sulfate groups of SDS. In addition, PEO is not expected to modify the negative charge of the aqueous pores. Consequently, the observed exclu- sion of the PEO-bound SDS micelies from the aqueous pores, coupled with the observed inclusion of the free SDS micelies into the aqueous pores, should result entirely from the proposed micelie steric hindrance mechanism embodied in penetration model (iii). Future work aimed at studying the effect of electrostatics on permeant penetration into the epidermis should consider the skin penetration of fixed-size charged species at different ionic strengths.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)