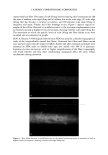
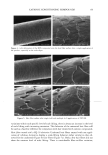
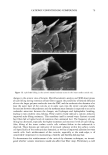
j. Cosmet. Sci., 54, 29-46 (January/February 2003) Challenging the surfactant monomer skin penetration model' Penetration of sodium dodecyl sulfate micelies into the epidermis PETER N. MOORE, SUDHAKAR PUVVADA, and DANIEL BLANKSCHTEIN, Department of Chemical Engineering, Massachusetts Institute of Technology, Cambridge, MA 02139 (P.N.M., D.B.), and Unilever Home and Personal Care NA, Trumbull, CT 06611 (S.P.). Accepted for publication November 15, 2002. Synopsis The penetration of sodium dodecyl sulfate (SDS) into the epidermis was measured using •4C-radiolabeled SDS. It was found that, at surfactant concentrations that exceed the critical micelle concentration (CMC) of SDS, the concentration of SDS measured in the epidermis increased as the total SDS concentration in the solution contacting the skin increased, thus demonstrating that miceIlar SDS contributes to the penetration of SDS into the epidermis. The observed SDS dose-dependent response contradicts the widely accepted view that only surfactant monomers penetrate into the skin, while surfactant in micellar form does not contribute to surfactant penetration into the skin. Nevertheless, this finding is consistent with previously unexplained observations of a dose-dependent damage to the skin induced by SDS at concentrations above the CMC. When poly(ethylene oxide) (PEO) was mixed with SDS, SDS micelles bound to PEO did not contribute to the concentration of SDS in the epidermis, while SDS in free SDS micelles did. Dynamic light-scattering measurements revealed an average hydrodynamic radius of 20 ]k for the SDS micelies, and a larger radius of 25 ]k for the PEO-bound SDS micelies. A comparison with typical aqueous pore radii in the stratum corneum measured in the literature (10-28 ]k) suggests that the SDS micelles may be able to penetrate into the skin, while the PEO-bound SDS micelles may be sterically hindered from penetrating into the skin. INTRODUCTION The investigation of surfactant-induced skin irritation has been extensive, including (i) which surfactants are most irritating (1-8), (ii) how mixing surfactants can reduce skin irritation (9-11), and (iii) how surfactants can lead to changes in the permeability of the skin (12-18). All the proposed mechanisms for surfactant-induced skin irritation involve the penetration of surfactants into the skin, where they can (i) denature proteins (2,4,19- 21), (ii) remove lipids from the stratum corneum (SC) (22-25), or (iii) disrupt the Address all correspondence to Daniel Blankschtein. 29
30 JOURNAL OF COSMETIC SCIENCE integrity of the lipid bilayers in the SC and the viable epidermis (12,16-18,26,27). The widely accepted view regarding surfactant-induced skin irritation is that, at surfactant concentrations that exceed the critical micelie concentration (CMC), where surfactant micelies first form, only surfactant monomers can penetrate into the skin, either because surfactant micelies are not surface-active, or because they are too large to penetrate into the SC (2,5,7,9,10,15,28,29). This view is based primarily on experimental observations using mixtures of surfactants, where surfactant-induced skin irritation was correlated with the CMC of the surfactant mixtures examined (5,9-11). This correlation is viewed as evidence that only the surfactant monomers are responsible for skin irritation, because the CMC is approximately equal to the surfactant monomer concentration. We call this widely accepted view the surfactant monomer skin penetration model and will refer to it hereafter as the monomer penetration model If surfactant-induced skin irritation is controlled solely by the monomeric surfactant, then it would naturally follow that there should be no effect of increasing the total surfactant concentration beyond the CMC, since the surfactant monomer concentration should remain approximately constant in that case. Instead, it has been observed ex- perimentally that as the total surfactant concentration is increased beyond the CMC, the surfactant-induced damage to the skin increases as well (1-3,7,10,13,15). For example, Agner and Serup found that the severity of the transepidermal water loss (TEWL) induced by the anionic surfactant SDS increased as the SDS concentration increased beyond the CMC of SDS (8.7 mM) (1). In other studies, it was found that as the SDS concentration increased beyond the CMC, the amount of SDS that penetrates into the stratum corneum also increased (2,28,29). Yet another illustration of the effect of increasing the total surfactant concentration above the CMC is provided by Rhein et aL (10). Specifically, these authors demonstrated that mixing SDS with a second, milder anionic surfactant, alkyl 7-ethoxy sulfate (AEOS- 7), led to a reduction in the irritation potential of SDS that correlated with a reduction in the CMC of the surfactant mixture, thus providing evidence for the monomer pen- etration model. However, the authors also noted that when the total surfactant concen- tration was increased, the irritation also increased, a finding that is not consistent with the monomer penetration model and that suggests a more complex mechanism of surfactant-induced skin irritation. Various explanations have been put forward to rationalize the observed dose-dependent effect of surfactants on the skin at surfactant concentrations above the CMC, including (i) that the monomer activity increases above the CMC (15) and (ii) that the micelies in the contacting solution solubilize lipids present in the skin (22,28). However, regarding explanation (i), various measurements, including surface tension and solution conduc- tivity, clearly indicate that the surfactant monomer activity remains fairly constant, or even decreases, as the surfactant concentration increases beyond the CMC (30,31). With regard to explanation (ii), there is evidence that micelies are able to remove some lipids from the skin, but the observed changes in the skin lipid concentration and composition are rather small and difficult to interpret (22-25). According to explanation (ii), an increase in the number of micelies above the CMC should lead to an increase in the lipid solubilization capacity of the contacting solution, thus explaining the increased damage to the skin, but not explaining the observed increased penetration of SDS into the stratum corneum observed by Faucher and Goddard (29), and by Ananthapadmanabhan et aL (28). In her comprehensive review of surfactant interactions with the skin, Rhein
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)