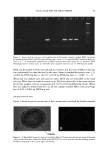
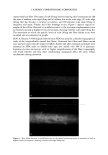
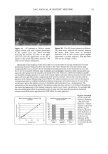
METHYLPARABEN IN O/W EMULSIONS 51 100 - 95 90 85 80 75 70 65 60 I I I I I 0.2 0.4 0.6 0.8 1 Sample Volume (mL) Figure 2. Effect of the sample volume on recovery of MP. Vertical bars represent SD. 1.145 1.14 1.135 1.13 1.125 1.12 1.115 1.11 100.0 100.5 101.0 101.5 102.0 102.5 103.0 103.5 Percent Theory Figure 3. Analysis of emulsions spiked with up to 3% additional MP.
52 JOURNAL OF COSMETIC SCIENCE ml was used. The average recovery from five samples was 92.0%, with a 0.7% CV. This data indicates that the assay method was reproducible. Precision and accuracy were evaluated by determining the MP concentration of six samples of the emulsion to which 0.5% to 3.0% additional MP was added in 0.5% increments. A linear relationship (R 2 = 0.9597) (Figure 3) between the theoretical MP concentration and the area ratio of the MP and BP peaks was obtained. In addition, analysis of a blank emulsion did not produce a peak corresponding to MP. Thus, it was concluded that the SPE extraction and HPLC analysis of MP in o/w emulsions is free of interference. CONCLUSIONS The use of the Oasis HLB cartridge (1 cc) effectively separated MP from other compo- nents of an o/w emulsion. This technique was simple, rapid, precise, and accurate, used a small volume of solvent, and required no drying stage. The determination of the amount of MP by SPE followed by HPLC analysis yielded well-resolved peaks of MP and the corresponding internal standard (BP) with excellent recovery (92.0%) and good precision (0.7%). ACKNOWLEDGMENTS This study was supported in part by the NSF Industry/University Cooperative Research Center in Pharmaceutical Processing. The authors gratefully acknowledge the technical assistance provided by Mr. Allen Anderson of Waters Corporation. REFERENCES (1) A. Wade and P.J. Weller, Handbook of Pharmaceutical Excipients, 2nd ed. (Pharmaceutical Press, London, 1994), p. 310. (2) D. Kollmorgen and B. Kraut,J. Chromatogr., B707, 181-187 (1998). (3) M. A. Abuirjeie, M. E. Abdel-Hamid, A. A. Abdel-Aziz, and E. A. Ibrahim, Anal. Letters, 23, 67-82 (1990). (4) L. Gagliardi, A. Amaro, A. Basili, G. Carazzutti, E. Gattavecchia, and D. Tonelli,J. Chromatogr., 315, 465•i69 (1984). (5) N. D. Brown, L. L. Hall, and H. K. Sleeman, J. Chromatogr., 166, 316-319 (1978). (6) F. A. Fitzpatrick, A. F. Summa, and A.D. Cooper, J. Soc. Cosmet. Chem., 26, 377-387 (1975). (7) Y. Maeda, M. Yamamoto, K. Owada, S. Sato, and T. Masui,J. Chromatogr., 410, 413-418 (1987). (8) U. Leuenberger, R. Gauch, and E. Baumgarmer, J. Chromatogr., 173, 343-348 (1979). (9) N. Pongcharoenkiat, G. Narsimhan, R. T. Lyons, and S. L. Hem, J. Pharm. Sci. 91,559-570 (2002).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)