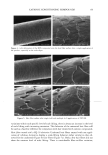
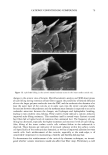
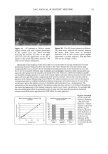
EPIDERMAL PENETRATION OF SDS MICELLES 43 CONCLUSIONS According ro a widely accepted view, suroeacranr micelies cannot penetrate into the skin, due ro size limitations or other oeacrors, and as a result, suroeacranr-induced skin irritation should be determined solely by the concentration ooe suroeacranr toohomers (2,5,7,9,10,15). •Vde have shown rhar this is nor the case for SDS. Instead, for a fixed amount ooe rime (five hours), we found rhar the amount ooe SDS rhar penetrates into the skin is directly related ro the concentration ooe SDS micelies present in the solution contacting the skin. •Vdhen PEO is added ro the SDS solution, SDS in micelies rhar are bound ro PEO does nor penetrate into the skin, while toohomeric SDS and SDS in free micelies do. A regression analysis of our experimental data indicated rhar the toohomeric SDS penetrates into the skin between two and three rimes oeasrer than the SDS in free micelies, while SDS bound ro PEO does nor penetrate into the skin significantly. However, ar the relatively high suroeacranr concentrations typically encountered in prac- tical applications, 5-10 wr% (where 1 wr% SDS corresponds ro 35 raM), the miceliar contribution will overwhelm rhar ooe the toohomers. A new model of suroeacranr penetration into the skin was required ro explain these results, since the current toohomer penetration model was unable ro do so. The model had ro be able ro explain both why SDS from free micelies is able ro penetrate into the skin, and why SDS from PEO-bound SDS micelies is unable to. •Vde proposed a new model ro explain these results, in which the free SDS micelies are able ro penetrate into the skin, while the PEO-bound SDS micelies are nor. SDS micelies are very hydrophilic, such rhar any penetration into the stratum comeurn will require a hydrophilic pathway. Such a pathway exists in the aqueous pores rhar are present in the stratum comeurn (53-56). In our new suroeacranr skin penetration model, the free SDS micelies can access the aqueous pores while the PEO-bound SDS micelies cannot. •Vde propose rhar the ability ro access the aqueous pores is determined by the size ooe the SDS micelie, or ooe the PEO-bound SDS micelie, relative ro the skin aqueous pore size. Several researchers have measured the average aqueous pore radius ooe the skin, and found ir ro be between l0 and 28 • (12,42-44). •Vde have measured the average radius ooe the SDS micelies, both in the presence and in the absence ooe PEO, and found rhar the free SDS micelies have a radius ooe about 20 •, while the PEO-bound SDS micelies have a radius ooe about 25 •. Therefore, the oeree SDS micelies are small enough ro access the aqueous pores, while the PEO-bound SDS micelies are srericMly hindered from pen- erraring into the aqueous pores of the skin. Although our results contradict the widely accepted view rhar suroeacranr in miceliar form does nor contribute ro suroeacranr penetration into the skin, there is ample evidence in the literature rhar the damage ro the skin is related ro the concentration ooe miceliar SDS in the contacting solution (1-3,7,10,13,15). •Vde have shown rhar this damage can be explained by the increased penetration ooe SDS into the skin, and we have also shown rhar this penetration can be reduced by changing the solution characteristics ooe the SDS micelies through the addition ooe PEO. Our findings and proposed new model ooe suroeacranr penetration into the skin should also be useful from a practical viewpoint, as they provide a new operarionM strategy for reducing the penetration ooe a known irritant (SDS) into the skin, which will hopefully reduce the damage caused by SDS through a reduction in its dose in the skin. In this new strategy, one must consider the possible contribution of the suroeacranr micelies, in
44 JOURNAL OF COSMETIC SCIENCE addition to the contribution of surfactant monomers, to the penetration of the surfactant into the skin. As stressed earlier, depending on the type of surfactant system contacting the skin, electrostatic effects between charged micelies and the charged aqueous pores may need to be considered, in addition to the micelie steric hindrance considerations put forward in this paper, to elucidate the ability of charged micelies to penetrate into the epidermis. In the future, we plan to examine how mixing surfactants affects surfactant penetration into the skin, since results reported in the past using mixed surfactant systems have been utilized as evidence to support the monomer penetration model (5,9-11), which we have shown not to be applicable in the case of SDS. We also plan to investigate the rela- tionship between the penetration of surfactant into the epidermis and the actual damage induced to the barrier properties of the skin. ACKNOWLEDGMENTS We thank Dr. Hua Tang for many useful discussions regarding the preparation of skin samples and other experimental details, as well as on hindered-transport theories. We also thank Dr. Roxanne Pouliot and Allison Fielder for conducting some of the experi- ments and testing the surfactant penetration method. Finally, we thank Dr. Linda Rhein for bringing reference 32 to our attention, as well as for illuminating discussions on surfactant-induced skin irritation. We are grateful to Unilever Home and Personal Care NA for financial support of this work. Peter Moore thanks the Natural Sciences and Engineering Research Council of Canada for the award of a PGS-B scholarship. REFERENCES (1) T. Agner and J. Serup, Sodium lauryl sulphate for irritant patch testing--A dose-response study using bioengineering methods for determination of skin irritation,J. Invest. Dermato/., 95,543-547 (1990). (2) J. A. Faucher and E. D. Goddard, Interaction of keratinous substrates with sodium lauryl sulfate. II. Permeation through stratum corneum, J. Soc Cosmet. Chem., 29, 339-352 (1978). (3) C. H. Lee and H. I. Maibach, Study of cumulative irritant contact dermatitis in man utilizing open application on subclinically irritated skin, Contact Dermatitis, 30, 271-275 (1994). (4) L. D. Rhein, C. R. Robbins, K. Fernee, and R. Cantore, Surfactant structure effects on swelling of isolated human stratum corneum, J. Soc Cosmet. Chem., 37, 125-139 (1986). (5) J. Vilaplana, J. M. Mascaro, C. Trullas, J. Coil, C. Romaguera, C. Zemba, and C. Pelejero, Human irritant response to different qualities and concentrations of cocoamidopropylbetaines: A possible model of paradoxical irritant response, Contact Dermatitis, 26, 289-294 (1992). (6) K.-P. Wilhelm, M. Samblebe, and C.-P. Siegers, Quantitative in vitro assessment of N-alkyl sulphate- induced cytotoxicity in human keratinocytes (HaCaT). Comparison with in vivo human irritation tests, Br. J. Dermato/., 130, 18-23 (1994). (7) K.-P. Wilhelm, A. B. Cua, H. H. Wolff, and H. I. Maibach, Surfactant-induced stratum corneum hydration in vivo: Prediction of the irritation potential in anionic surfactants, J. Invest. Dermatol., 101, 310-315 (1993). (8) K.-P. Wilhelm, G. Freitag, and H. H. Wolff, Surfactant-induced skin irritation and skin repair,J. Am. Acad. Dermato/., 30, 944-949 (1994). (9) T.J. Hall-Manning, G. H. Holland, G. Rennie, P. Revell, J. Hines, M.D. Barratt, and D. A. Bas- ketter, Skin irritation potential of mixed surfactant systems, Fd. Chem. Toxic, 36, 233-238 (1998). (10) L. D. Rhein, F. A. Simion, R. L. Hill, R. H. Cagan, J. Mattai, and H. I. Maibach, Human cutaneous
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)