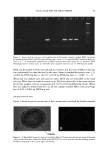
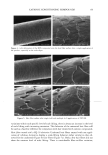
CATIONIC CONDITIONING COMPOUNDS 67 and styling products, because of their beneficial effects in grooming and in the appear- ance of the hair assembly. Cationics display a high degree of affinity to keratin. Hair fibers have a low isoelectric point, which is near pH 3.67 (2) in unaltered human hair and even lower in bleached hair. Therefore, hair fibers are negatively charged at any pH above this point, and since cosmetic treatments are normally carried out above this pH value, the cationic polymers are readily adsorbed by keratin. Besides their own affinity to hair, cationic polymers can also cause other components of the formulations into which they are incorporated to adhere to keratin. This is a property that is utilized by many products, such as the "leave-on" products and rinses. Interaction between cationics and keratin is believed to occur mainly at the hair fiber surface, and only low-molecular-weight (-1000 Da) conditioners are expected to diffuse deeper into the hair fiber interior, most likely by way of intercellular diffusion. High- molecular-weight cationics are not expected to significantly penetrate, except for a short distance into the CMC and possibly the endocuticular domains of the outer layer of the cuticula. Low-molecular-weight conditioners were shown to adsorb faster and to a greater extent (due to their ability to diffuse into the hair shaft) than the highest- molecular-weight polymers, which show a rapid initial uptake followed by leveling, indicating saturation of the hair fiber surface by the polymer and limited penetration, if any (3). The cationics restricted to deposition at scale edges and on scale faces are expected to 60•038 5.0 kV Figure 2. Continued.
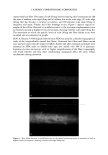
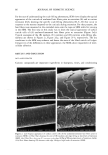
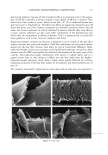
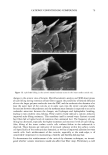
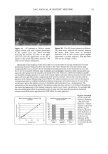
68 JOURNAL OF COSMETIC SCIENCE perform differently during hair fiber extension than those capable of either diffusing into the hair fiber interior or achieving both surface deposition and penetration into the cuticular sheath. Our SEM studies had shown significant adsorption of the polymeric cellulose and guar derivatives onto the hair fiber surface, especially at the scale edges, with little or no additional uptake after multiple applications of these two polymers. This appears to indicate saturation of available anionic sites on the hair fiber surface after the first application. Micrographs of typical sorption of the celldose and guar derivatives onto the hair fiber surface are shown in Figures 3a,b and 4a,b, respectively, displaying preferential deposition at the scale edges. The low-molecular-weight CETAB, on the other hand, appears to behave differently from the polymeric materials. After a single application of this compound, the hair fiber surface appears rather "clean" and free of deposits (Figure 5a,b). After multiple appli- cations of CETAB, the surface of many hair fibers still appears rather "clean" (Figure 5c), while other fibers clearly show thin film-like deposits on the scale faces and edges and leave no doubt about the nature of the deposits (Figure 5d). MICROFLUOROMETRIC STUDY OF THE EFFECTS OF MCCs ON SCALE LIFTING To determine the effects of single and multiple applications of cationics on the scale- lifting phenomenon of the surface cuticle cell, hair fibers have been extended up to failure while being observed in autofluorescence in the fluorescence microspectropho- tometer. The extensions at which specific levels of scale lifting and fiber failure occur were recorded and plotted. It should be pointed out that the break extensions observed in this work are different from those observed in a regular tensile test, which is carried out at a higher extension rate (-50%/min.). Break extensions are higher in this case, because of extension of the hair in steps, which allows the fiber to relax between steps. Unaltered hair without MCC treatment. Surface cuticle cells of untreated, unaltered hair fibers tend to respond with various levels of scale lifting to release the stresses of extension, until finally hair failure occurs (Figure 6). Even though there are fiber-to-fiber Figure 3. (a,b) Adsorption of the PQ-10 compound onto the hair fiber surface after a single application of the product, especially at the scale edges.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)