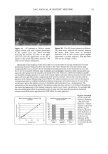
88 JOURNAL OF COSMETIC SCIENCE Results and Discussion The dielectric constants of the individual chemicals in this study, as well as other chemicals commonly found in sunscreens, can be found in Table I. The largest number of formulations tested in this study contained, as UV filters, octyl salicylate (5%), oxybenzone (3%), and avobenzone (2%). The compositions of the formulations' oil phases and the results of the measurements made on these formulations are reported in Table II and, graphically, in Figure I. The results of in vivo studies on four of the formulations are also reported in Table IL The study's early results seemed consistent with a logarithmic relationship between oil phase dielectric constant and rate ofphotodecay (i.e., as the dielectric constant increases, the photodecay rate decreases and the sunscreen becomes more photostable). However, as more data became available, regression analysis determined the best curve fit to be parabolic (i.e. as the dielectric constant increases, the photodecay rate decreases up to a point beyond which it increases.) Other filter systems tested produced confirmatory results. A parabolic relationship between the dielectric constant of the oil phase and the photostability of an avobenzone-containing sunscreen is reminiscent of the theories of Rudolph A. Marcus of the California Institute of Technology. In a series of papers written in the late Fifties and early Sixties, Marcus proposed that the solvent mediates electron transfer reactions and the rate of electron transfer is related to the driving force by a quadratic expression that is descriptive of a parabola? In 1992, Marcus was awarded the Nobel Prize in Chemistry for his contributions. The similarity of our findings to Marcus Theory is perhaps not surprising given the prominent role played by charge transfer in the causation and amelioration of photodecay. Conclusion In many sunscreens, the photodecay rate, k•,d, is related to the polarity of the sunscreen oil phase by the expression, ae: + be + c, where e is the dielectric constant of the oil phase (comprising the solvents, emollients and UV filters), and a, b, and c are empirically derived values for a given filter combination. The expression describes a parabola, the vertex of which identifies the dielectric constant at which photodecay will be minimized and, therefore, sunscreen performance may be optimized. 0 -0.015 -0.03 -0.045 -0.06 -0.075 -0.09 -0.105 -0.12 -0.135 -0.15 Dielectric Constant vs. Photodecay kpd "-.004215s 2 + 0.072748s - 0.33701 Vertex=8.63 R 2 = 0.992 ß Dielectric Constant Figure I Photodecay rate constants of formulations containing, as UV filters, octyl salicylate (5%), oxybenzone (3%), avobenzone (2%) plotted against the dielectric constants of their oil phases. The larger dots represent formulations for which in vivo (five subject, static) SPF studies were completed. Starting from the left, these formulations achieved SPF 17, 20, 21, and 25 respectively.
2002 ANNUAL SCIENTIFIC MEETING 89 Table I: Dielectric Constants of Common Lipophilic Sunscreen Ingredients (Ingredients listed in BOLDFACE t7pc were used in the study.) Chemical Isoeicosane Polyisobutene Mincral oil Octyldodecyl neopentanoate Ethylhcx¾1 stearate Eth¾1hcxyl palmirate Isopropyl palmirate lsopropyl myristate Ci 2-15 alk•l benzoates Caprylic/Capric tri$1¾cerides Dicth¾1hex¾1 adipate Dibutyloct•l malate But•loctyl salicylate Diethylhexyl malate Octyl methoxycinnamate Octyl salicylate Diisoamyl malate Avobenzone lsododecyl alcohol dimer with IPDI Dimethyl capramide Oxybenzonc Dielectric Constant •, 25øC 2.04 2.13 2.13 3.04 3.05 3.06 3.20 3.24 3.78 3.83 4.21 4.65 5.35 5.88 5.98 6.20 7.42 io (est.) 10 (est) i 2.43 i 3.0 (literature reference) Table II: Oil Phase Compositions, Measurements, and Results for Test Formulations Containin, 'late, 3% Ox•'benzone, 2% Avobenzone Ingredients A - B (5 D E F G H I Isopropyl myristate 10% C12-15 alkyl benzoates 10% 10% Diethylhexyl malate 10% 15¾o 10% 7.5% Saturated hydrocarbon 5% 5% 5% C20 neopentanoate 5% Diisoamyl realate 10% 9.25% Dibutyloctyl malate 5% 4% Dimethyl capram/de 1.75% 7.5% C12 dimer with IPDI 5% Dielectric constant 4.57 4.92 5.48 5.89 7.44 7.66 7.83 8.71 10.59 Photodecay rate constant -.095 -.078 -.062 -.055 -.028 -.027 -.024 -.015 -.032 In vivo SPF 17 20 21 25 i Turro, N.J., Modern Molecular Photochemist•7, Menlo Park, Benjam/n/Cummings, 1978, p. 329 2 Marcus, R.A.d. chem. Phys. 1956, 24, 966. Marcus, R.A. Annu. Rev. Phys. Chem. 1964, 15, 155.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)