96 JOURNAL OF COSMETIC SCIENCE IDENTIFICATION AND INVESTIGATION OF POTENTIAL FRAGRANCE- RETAINING MATERIALS USING ANALYTICAL AND SENSORY METHODS Regina M. Malczewski, Ph.D. and Deborah L. Meyers, Ph.D. Dow Corning Corporation Introduction The total US fragrance market in 2001 reached sales of $5 billion (1) the prestige portion of the market accounted for $2.9 billion (2). Market research has shown that among fragrance users, 53% of the women and 50% of the men are dissatisfied with some aspect of fragrance performance. Those surveyed complained that fragrances fade too rapidly, distort over time, or cause allergic reactions (2). Therefore, materials that enhance retention of a given concentration of fragrance would improve perception as well as allow the use of lower levels to decrease allergenicity and cost. Methods to measure fragrance retention can be instrument-based (gas chromatography) or sensory-based (human panel) this paper compares both methods and discusses the results obtained with each. Materials and Methods A. Analytical method A Franz diffusion cell apparatus (Fig 1, Crown glass) was used to evaluate the ability of silicones to slow the release of fragrance from a test mixture of four fragrances. Each two-part glass cell was composed of a top that contained a chamber with charcoal (for trapping volatiles SKC) and a matched bottom. The test fragrance mixture was placed on a glass membrane (7.1 cm 2, Osmonics) that was sealed in place with a Teflon-coated O-ring that nested into both the top and bottom pieces. Each cell was water- jacketed at the bottom (35øC) air entering at a sidearm circulated through the chamber and trap at 145 cc/min. Depending on the viscosity of the test mixture, 250 pl was applied to the filter by positive displacement pipette (Gilson), or -250 pg was applied by spatula. The composition of the fragrance mixture, in order of decreasing volatility, was as follows: isoamyl acetate, eucalyptol, methyl benzoate, and geraniol (Aldrich, all 96% pure). A 1:1:1:1 combination of each fragrance was used as 5% of the test sample. The remainder of the mixture was silicone, which varied from dimethicone (non-volatile, low viscosity negative control) to new materials (A, B, and C) that were 12- 15% non-volatile content. Four hr after applying mixture and completing set assembly, the charcoal from each trap was retrieved and extracted using toluene (Aldrich, HPLC Grade). The extract was analyzed by GC (HP 6890, FID, DB- 1 column) using isododecane as an internal standard. Analysis of Variance (JMP SAS Institute, v 4.0) and Tukey's HSD test (significance level set at c•=0.05) were used to establish meaningful results. B. Sensory method A panel of volunteers trained to recognize, rank, match and name simple fragrances was used to examine the potential of various silicones to retain fragrances over time periods of up to 16 hr. Aloe Lily (AL) and Powder Fresh (PF), common fragrance types used in personal care, were mixed at levels from 0.5 to 1% in prototype water-in-silicone or water-in-oil formulations (Tables 1 and 2). Silicones of interest (12- 15% non-volatile content) were present in the test mixtures at levels between 2 and 5% active. Samples (0.05g) were spread into the bottoms of polystyrene dishes of 2" in diameter (Pall Gelman Laboratory). The dishes (with fitted lids) were then stacked and placed at 4øC until opened at RT for the designated "exposure time" (1-15 hr). After exposure was complete, the plates were closed and random-ordered sets were prepared for panelist evaluation (M hr later). Phase 1 of the assay involved evaluation of each formulation at all time points so the panelists could rank the intensity over time for each sample separately. The second phase involved the evaluation of all formulations against each other at each time point so that a comparison of the efficacies of the formulations could be made. The panelists were instructed to rank order the plates in each set from weakest fragrance to strongest. A Friedman's Rank test and a non-parametric analogue of Fisher's Least Significant Difference Test (3) , as well as the binomial distribution with a significance criterion of 0.9, were used to determine meaningful results in the sensory assay. Results and Discussion The analytical method showed significant differences between dimethicone and several experimental silicones (see Figs 3 and 4, for an example). The flexibility of this method is limited due to the time required to apply thick materials as a paste (and the resulting loss of volatiles and inaccuracy of results. Analytical testing was limited to four hours for screening purposes and practical reasons.
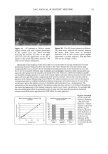
2002 ANNUAL SCIENTIFIC MEETING 97 Sensory testing can be done easily on actual formulations over whatever time frames are of interest. Phase 1 fragrance results give information about fragrance release and panel capability. Phase 2 panel assay results can determine the impact of parameters such as fragrance (Fig 3) and formulation (Fig 4) types on release kinetics, and have shown that a number of silicones have potential fragrance retention benefits. Reproducibility was demonstrated (data not shown), but interpretations are sample-set dependent. Conclusions Analytical and sensory methods both suggest the same silicones may sustain fragrance release, but the sensory method is more relevant, more flexible, and less complicated. References 1. Pringle, L. Explore Your Senses: Smell Benchmark Books, Marshall Cavendish, NY 2000 2. Marchie, M., "A Fresh Start for Fragrance" HAPP1, April, 2002 http://www. happi.com/current/Apri1022.htm 3. Meilgaard, M, Civille, G.V., and Cart, B. T., Sensory Evaluation Techniques, 3 ra Edition, CRC Press, Boca Raton, pg. 292, 1999 FIGURE 1: Individual Diffusion Cell mP• FIGURE 2: Fragrance Retention by Experimental Silicones Analytical Fragrance Results Colored bare are aig diff from control at alpha=O.05 6OO 300 15g Fragrance Component al Dimethicone, 10 cs [] Silicone A El Silicone B, hi DP FIGURE 3: Sensory Testing Results, Silicone A Panel Test I•sults, Fragrance Impact Stippled bar is sig dlff from control by binomial distribution 50 40 2O 10 0 Fragrance formulation B control [] 5% Sil A wlAL, 15 hr I•$%S Aw/PF, 10 hr TABLE l: Water-in-Silicone Formulation INGREDIENT WT % ,,, Glycerin 5 Sodium Chloride 1 Cyclopentasiloxane 10 Cyclopentasiloxane (and) 10 PEG/PPG-18/18 Dimethicone Silicone X 2-5 Fragrance 0.5 - 1 FIGURE 4: Sensory Testing Results, Silicone B Panel Results, Formulation Impact Checkered bar is alg diff from control at alpha =0.05 TABLE 2: Water-in Oil Formulation INGREDIENT WT % Carbomer (1% soln) 8.5 Water q.s. Steareth-2 2 isododecane i0 Silicone X 2-3 Fragrance 0.5-1
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)