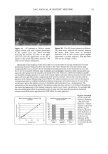
EPIDERMAL PENETRATION OF SDS MICELLES 39 The regression analysis yielded a value of c that is two orders of magnitude smaller than that of a or b, and was not significantly different from zero. Accordingly, a second regression analysis was performed where c was set equal to zero. Removing the c pa- rameter from the regression analysis led to a smaller confidence interval for the value of a, while the regressed value of b remained the same. The second regression analysis yielded: a = 0.14 + 0.04 C,•,/C ........ b = 0.043 + 0.006 Cskir•/Cj•e e mic'eZZes The important conclusion that can be drawn from the regression analysis is that SDS in free micelies contributes to the amount of SDS measured in the epidermis, while SDS in PEO-bound micelies does not. The regression analysis also shows that the contribution of free SDS micelies to C,•i, is independent of the presence of PEO, because the value of the parameter b is the same whether or not the data with PEO is included. When one considers that in surfactant-containing washing formulations the concentration of mi- celies is typically much higher than that of the monomers, it is evident that, with regard to the penetration of surfactant into the skin, it is important to consider the contribution of the micelies in addition to that of the monomers. DYNAMIC LIGHT-SCATTERING RESULTS Figure 6 shows the results of the DLS measurements, with the micelie size being determined by extrapolation to a zero micelie concentration, which occurs at the CMC (about 0.9 mM SDS for free SDS micelies) or at the CAC (about 0.6 mM SDS for PEO-bound SDS micelies) of the surfactant solution. Using a linear regression analysis, we have determined that the average radius of the free SDS micelies is 20/t and that the PEO-bound SDS micelies have an average radius of 25 •. 30 e 25 o 20 15 0 10 20 30 40 Concentration of Miceliar SDS (mM) Figure 6. Measured effective radii of SDS micelies in the absence (O) and in the presence ([•) of PEO plotted versus the SDS concentration minus the CMC, or the SDS concentration minus the CAC, respec- tively, using DLS measurements at 25øC. The miceliar radii were determined using a CONTIN analysis. The error bars reflect a 95% confidence interval based on eight samples at each SDS concentration.
40 JOURNAL OF COSMETIC SCIENCE NEW PROPOSED MODELS OF SURFACTANT PENETRATION INTO THE EPIDERMIS We have demonstrated that the amount of SDS that penetrates into the epidermis is determined by both the concentrations of SDS monomers and of free SDS micelies in the contacting solution, and that the concentration of SDS micelies bound to PEO has no effect on SDS penetration into the skin. Because, as we have shown, the monomer penetration model cannot explain the experimental observations in Figures 1, 2 and 5, we next attempted to identify a surfactant skin penetration model that would be con- sistent with our experimental observations. We believe that the key to the new surfac- tant skin penetration model lies in the different behaviors observed for SDS in free micelles and in SDS micelies bound to PEO, since the former contribute to surfactant penetration into the epidermis while the latter do not contribute, as shown by the regression analysis. Below, we discuss three possible surfactant skin penetration models to explain our experimental observations: (i) micelie kinetics affect the rate of surfactant monomer replenishment, (ii) micelies disintegrate when they approach the skin, and (iii) micelies can penetrate into the skin, but there is a size limitation. Penetration model (i) utilizes the kinetics of micelie dissolution (48,49) to explain the observed increase in SDS penetration into the epidermis with increasing SDS concen- tration in the contacting solution, and is an extension of the original monomer pen- etration model. In this model, as the SDS monomers in the contacting solution penetrate into the SC, they must be replenished, either by the breakup of SDS micelies into monomers or by the diffusion of other SDS monomers present in the contacting solution farther away from the SC. As the SDS micelie concentration in the contacting solution increases, the argument goes, the rate of monomer formation through micelie dissolution should increase, and hence the concentration of SDS monomers adjacent to the SC should be higher in the presence of more SDS micelies. As appealing as this model appears initially, it fails in several respects. First, it is well known that as the SDS concentration in pure water increases from 50 to 200 mM, the rate of monomer formation through micelie dissolution actually decreases, with the SDS micelies at 200 mM being the most stable (50). Therefore, as the concentration of SDS in the contacting solution is increased, the rate of SDS monomer formation should decrease. According to this kinetic model, the SDS penetration into the epidermis should actually decrease, because the concen- tration of SDS monomers adjacent to the SC will be lower as the concentration of SDS in the contacting solution increases. A typical time constant for the breakup of SDS micelles to replenish the decreased SDS monomer supply, as SDS molecules penetrate into the skin, is between 0.01 s and 1 s (50). The characteristic time constant for diffusion across a membrane is described by L2/D, where L is the membrane thickness and D is the rate of diffusion through the membrane. The SC is about 15-pm thick, and the diffusion of surfactants across the SC is at least 1000 times slower than in bulk water (2), or approximately 10-•3 m2/s. This yields a characteristic diffusion time constant of about 2000 s, several orders of mag- nitude slower than the replenishment rate of the SDS monomers as SDS molecules penetrate into the SC. This simple timescale analysis suggests that the rate-determining step of diffusion into the skin is governed by the diffusion through the stratum corneum, and not by the micelie kinetics. Another problem with the micelie-kinetic model is that the addition of PEO to a SDS solution has been shown to increase the rate of micelie dissolution dramatically (51,52).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)