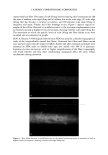
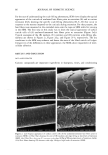
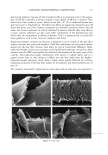
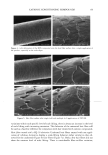
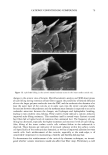
22 JOURNAL OF COSMETIC SCIENCE In the present study, procedures were established to extract DNA from hair fibers, to confirm that extracted DNA was of human origin using the Pico-green © reagent (PCR), and to quantify DNA in extracts. Sodium dodecyl sulfate (SDS) washes have been shown to reduce the level of DNA in hair (5). Experiments were therefore carried out to investigate the effects of common basic shampoo on the level of DNA in hair. DNA has been isolated specifically from hair cuticle (2), suggesting that this may be the location of DNA within the hair shaft. Experiments were therefore also carried out to investigate the location of DNA within the shaft using DNA-specific fluorescent stains and fluorescence microscopy. METHODS EXTRACTION OF DNA FROM HAIR SHAFTS Freshly clipped hair was washed in ethanol for 5 min and rinsed in distilled water before being cut as finely as possible with hair scissors, and digested overnight at 56øC in 0.01 M Tris-HCl (pH 8), 0.005 M EDTA, 0.1 M NaC1, 0.039 M DTT, 2% SDS, and 250 lag of proteinase K (Gibco) per ml of extraction buffer. The digested hair solution was then extracted twice with two volumes of phenol satu- rated by Tris-EDTA (TE) buffer (Sigma), and once with two volumes of chloroform. The solutions were centrifuged between each extraction. The resulting DNA in the final aqueous phase was then precipitated using two and a half volumes of ice-cold absolute ethanol. The DNA was pelleted by centrifugation, and the resulting pellet washed in 70% ethanol and centrifuged for a further 10 min. The DNA pellet was air-dried for 20 min before being resuspended in 100 lal of TE buffer for the Pico-green © assay (Mo- lecular Probes), or 15 lal for PCR. The method was based on that previously described by Uchihi et •L (2). Gloves were used at all times to prevent DNA from the hands contaminating samples. IDENTIFICATION OF EXTRACTED DNA FROM HAIR SHAFTS The concentration of DNA was determined using the Pico-green © reagent, which is a fluorescent nucleic acid stain for specifically quantitating double-stranded DNA. Vari- ous amounts of clipped hair were used from one hair (approximately 1 mg) up to 200 mg of hair. The unknown concentrations of DNA were determined from extracted hair samples by using a spectrofluorimeter (Spex Fluoromax) according to the manufacturers instruc- tions, employing human genomic DNA for calibrations (Promega), and were calculated in relation to the amount of starting material (ng of DNA per mg of hair). PCR AMPLIFICATION OF THE HLA-DQA1 GENE PCR reactions consisted of 1 x PCR buffer (10 mM of Tris-HC1 [pH 8.3], 500 mM of KCI), 1.5 mM of MgCl2, 200 laM of each dNTP (dCTP, dGTP, dATP, dTTP), 50 pmol of each primer, and 1.25 units of Amplitaq in a volume of 25 lal. The primers designed
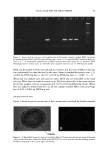
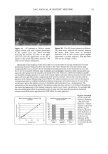
DNA ANALYSIS OF HAIR FIBERS 23 for amplification of the HLA-DQA 1 genotype were (5'-3'): GGT GTA AAC TTG TAC CAG and GGT AGC AGC GGT AGA GTT G (1). Thirty cycles were carried out, consisting of denaturation at 94øC for 1 min, annealing at 54øC for 1 min, and extension at 72øC for 1 min, before a final extension step of 5 min at 72øC. Fifteen microliters of extracted hair DNA was used for amplification. To attempt to remove water-soluble melanins, some DNA samples were purified using Wizard DNA cleanup columns (Promega), according to the manufacturers instructions, before use in the PCR reaction. The resulting amplified samples were loaded on a 1.5% agarose gel, and run for 1 h at 100 V, stained with ethidium bromide, and photographed on a UV transilluminator. EFFECTS OF COLORING ON LEVELS OF HAIR SHAFT DNA Female subjects planning to have their hair permanently colored for the first time were recruited from the laboratory. Subjects had samples of hair taken before the hair treat- ment was carried out, and they returned to give a second sample after they had had their hair colored. DNA was extracted from the exact same amount of hair from each before-and-after treatment sample (around 100 mg), and the concentrations of DNA were determined by using the Pico-green © assay and related back to the exact amount of starting material. DNA LEVELS IN ROOT-END VERSUS TIP-END OF HAIR SHAFTS Root-end and tip-end hair was separated from 1-g Spanish hair switches (International Hair Importers). DNA was extracted from the same amount of hair for root and tip-end hair (around 100 mg), and the concentrations of DNA were determined by using the Pico-green © assay and related back to the exact amount of starting material. EFFECTS OF SHAMPOO WASHING ON DNA LEVELS OF HAIR SHAFTS Four samples of 50 mg of finely cut root-end switch hair were washed once for 30 sec in 1 ml of a 1:10 dilution of 12:2 base shampoo (12% sodium lauryl ethyl sulfate, 2% tegobetaine), and the wash solution was removed for purification and quantitation of DNA. Four samples of 50 mg of finely cut root-end switch hair were washed five, ten, or 20 times for 30 sec each time in 1 ml of a 1:10 dilution of 12:2 base, with 30-sec rinses in distilled water between each wash. The washes were pooled, and a 1 ml solution was removed for purification and quantitation of DNA. Four samples of 100 mg of finely cut switch hair was soaked in 2 ml of a 1:10 dilution of 12:2 shampoo base for 2 h. The wash solution was removed for purification and quantitation of DNA. The DNA concentrations were determined using the Pico-green © reagent, and the total amount of DNA lost during each washing procedure was calculated.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)