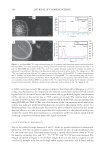
JOURNAL OF COSMETIC SCIENCE 320 non-lotioned and three lotioned products. For each product, 80 to 130 data points were collected from about 40 subjects. Large data sets were needed because some of the mar- keted lotioned products had a wide variation in the level of lotion on the tissue. These results are summarized in Table II. The relatively high standard deviations refl ect the total variation of lotion level within the tissue products (i.e., sheet-to-sheet difference), the variability in delivery of lotion from the tissue to the skin, possible skin lipid changes caused by mechanical action on the skin, and variation in the measurement. However, the measurement itself is quite good, as shown by the calibration curve linearity and high correlation (R2 0.99). Product 3 has the highest relative standard deviation (CV) of lotion delivered to the skin due to the high variation in lotion level on the tissue (due to machine variation in lotion applica- tion). In this case, we measured bulk lotion concentrations ranging from 3.99 to 5.77 g lotion/m2 subsrate. This high variation, from tissue to tissue, and the destructive nature of the bulk lotion concentration method (solvent extraction), prevented us from measur- ing the lotion transfer effi ciency (% transfer) for individual tissues. The wide variation in lotion amount, nearly 50%, on a marketed facial tissue (i.e., Product 3), necessitated a large sample size thus 80 to 130 data points per product were collected. Moreover, given the wide variation in lotion amount on each marketed product, one should expect a wide standard deviation in the amount of lotion transferred to the skin. Thus, in studies where the test products have a smaller variation in their lotion amount, a much smaller statistical variation in lotion transfer to the skin should be expected. CONCLUSIONS As expected, the two non-lotioned products delivered no lotion to the skin. The standard deviations may indicate surface changes to lipids that affected the spectrum to a small extent. The differences in lotion delivery to the arm are due to tissue lotion levels and lotion formulation differences. Product 3 delivered much less lotion to the skin than Products 4 and 5. The in vivo skin analysis described here is signifi cantly faster, simpler, and less invasive to the body than the tape stripping method described in the literature (1–3). INSA’s ability to quantify the deposition of compounds such as lotions onto human skin by products such as substrates and tonics, using ATR FT-IR, has been described. INSA’s utility was especially useful due to being noninvasive and nondestructive, which allowed Table II Average μg Lotion Deposited per cm2 of Volar Forearm Skin μg Lotion deposited per cm2 Non-lotioned tissue (Product 1) Non-lotioned tissue (Product 2) Lotioned tissue (Product 3) Lotioned tissue (Product 4) Lotioned tissue (Product 5) Average deposited -0.02 0.18 3.66 17.11 15.97 Standard deviation ±2.62 ±1.83 ±4.81 ±12.95 ±10.28 Relative standard deviation — — 131% 76% 64%
LOTION TRANSFER TO SKIN 321 us to study lotion deposition accurately, inexpensively, and quickly without compromis- ing our human subjects’ skin. ACKNOWLEDGMENTS Brooke Crase Woods and Phil Ritter assisted with the lab work. Melanie Hansmann assisted with the human subject protocol. REFERENCES (1) C. Surber, F. P. Schwab, and E. W. Smith, “Tape-Stripping Technique,” in Percutaneous Absorption-Drug- Cosmetics-Mechanisms-Methodology, 3rd ed., H. Bronaugh and H. Maibach, Eds. (Marcel Dekker, New York, 1999), pp. 395–409. (2) P. Lampen, W. Pittermann, H. M. Heise, M. Schmitt, H. Jungmann, and M. Kietzmann, Penetration studies of vitamin E acetate applied from cosmetic formulations to the stratum corneum of an in vitro model using quantifi cation by tape stripping, UV spectroscopy, and HPLC, J. Cosmet. Sci., 54, 119–131 (2003). (3) J. Lademann, A. Ilgevicius, O. Zurbau, H. D. Liess, S. Schanzer, H. J. Weigmann, C. Antoniou, R. v. Pelchrzim, and W. Sterry, Penetration studies of topically applied substances: Optical determination of the amount of stratum corneum removed by tape stripping, J. Biomed. Opt., 11(5), 54026-1–54026-6 (2006). (4) N. J. Harrick, Internal Refl ection Spectroscopy (Harrick Scientifi c Corp., Pleasantville, NY, 1979). (5) F. M. Mirabella, Jr., and N. J. Harrick, Internal Refl ection Spectroscopy: Review and Supplement (Harrick Scientifi c Corp., Pleasantville, NY, 1985). (6) V. H. W. Mak, R. O. Potts, and R. H. Guy, Oleic acid concentration and effect in human stratum cor- neum: Non-invasive determination by attenuated total refl ectance infrared spectroscopy in vivo, J. Con- trolled Release, 12, 67–75 (1990).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)