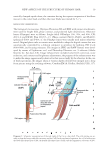
JOURNAL OF COSMETIC SCIENCE 28 CONCLUSIONS The structure of the hair cells and the fi ber shaft itself is substantially revised by optical microscopic observations of the hair components. Particularly, irrespective of the hair sources, that a mature Cu has a trowel-like shape with the following distinctive features: the part with handle-like shaped part (CuH) is full of Mf and usually colored by melanin granules, while the blade-like shaped parts fuses partially to build the honeycomb-like structure and a large cuticular thin plate. The medulla is tubular, streaming through all the hair shafts examined. The physicochemical stability of hair is mainly attributed to the presence of the honeycomb-like structure, the cuticular thin plate and the Mf of the Co and Cu. The present discoveries might be useful for people, especially chemists in the hair-related fi elds, to get new insights for their studies. ACKNOWLEDGMENTS We are deeply indebted to the staff members of Department of Applied Biochemistry, Osaka University and Department of Biochemistry, Osaka Municipal Technical Research Institute for many invaluable discussions and technical assistance throughout this work. We also thank Alan Hadley for permission to use the CombineZM software. REFERENCES (1) C. R. Robbins, Chemical and Physical Behavior of Human Hair, 5th ed. (Springer-Verlag, Berlin, 2012), Chapters 1 and 2. (2) C. E. Orfanos, W. Montagna, and G. Stüttgen (Eds.), Hair Research: Status and Future Aspects (Springer- Verlag, Berlin, 1981), Part I, pp. 3–234. (3) L.N. Jones, D. E. Rivett, and D. J. Tucker, “Wool and Related Mammalian Fibers” in Handbook of Fiber Chemistry, 3rd ed., M. Lewin Ed. (CRC Press, Boca Raton, FL, 2007), pp. 331–381. (4) K. Morioka, Hair Follicle, Differentiation under the Electron Microscope (Springer-Verlag, Tokyo, 2005). (5) L. J. Wolfram and M. K. O. Lindemann, Some observations on the hair cuticle, J. Soc Cosmet Chem, 22, 839–850 (1971). (6) J. A. Swift and B. Bews, The chemistry of human hair cuticle-I: A new method for physical isolation of cuticle, J. Soc. Cosm. Chem., 25, 13–22 (1974). (7) J. A. Swift and B. Bews, The chemistry of human hair cuticle-II: The isolation and amino acid analysis of the cell membranes and A-layer, J. Soc. Cosm. Chem., 25, 355–366 (1974). (8) J. A. Swift and B. Bews, The chemistry of human hair cuticle-III: The isolation and amino acid analysis of various subfractions of cuticle obtained by pronase and trypsin digestion, J. Soc. Cosm. Chem., 27, 289–300 (1976). (9) P. Kassenbeck, “Morphology and Fine Structure of Hair,” in Hair Research Status and Future Aspects, C. E. Orfanos, W. Montagna, and G. Stüttgen. Eds. (Springer-Verlag, Berlin, 1981), pp. 52–64. (10) R. H. Rice, V. J. Wong, and K. E. Pinkerton, Ultrastructural visualization of cross-linked protein fea- tures in epidermal appendages, J. Cell Sci., 107, 1985–1992 (1994). (11) R. H. Rice, V. J. Wong, K. E. Pinkerton, and J. P. Sundberg, Cross-linked features of mouse pelage hair resistant to detergent extraction, Ana. Rec., 254, 231–237 (1999). (12) Ta. Takizawa, To. Takizawa, S. Arai, M. Osumi, and T. Sato, Ultrastructure of human scalp hair shafts as revealed by freeze-substitution fi xation, Ana. Rec., 251, 406–413 (1998). (13) R. D. B. Fraser, T. P. MacRae, and G. E. Rogers, Keratins, Their Composition, Structure and Biosynthesis (Charles Thomas, Springfi eld, 1972), p. 56–74. (14) S. Ogawa, Y. Takeda, K. Kaneyama, and T. Komoto, Electron microscopic study of permanent straight- ening hair prepared by a reduction and heat treatment process, Sen’i Gakkaishi, 64, 352–357 (2008). (15) L. N. Jones, D. J. Peet, D. M. Danks, A. P. Negri, and D. E. Rivett, Hairs from patients with maple syrup urine disease show a structural defect in the fi ber cuticle, J. Invest, Dermatol., 106, 461–464 (1996).
NEW ASPECTS OF THE STRUCTURE OF HUMAN HAIR 29 (16) Biological Stain Commission (U.S.A.), “Staining Procedures,” 4th ed., G. Clark. Ed. (Lippincott Williams and Wilkins, Baltimore, 1981). (17) The image stacking software by Alan Hadley, was accessed in September, 2009 from http://www. hadleyweb.pwp.blueyonder.co.uk/ (18) J. A. Swift, “Morphology and histochemistry of human hair,” in Formation and Structure of Hyman Hair, P. Jolles, H. Zahn, and H. Höcker. Eds. (Birkhäuser Verlag, Basel, 1997), pp. 149–175. (19) W. G. Bryson, D. P. Harland, J. P. Caldwell, J. A. Vernon, R. J. Walls, J. L. Woods, S. Nagase, T. Itou, and K. Koike, Cortical cell types and intermediate fi lament arrangement correlate with fi ber curvature in Japanese human hair, J. Struct. Biol., 166, 46–58 (2009). (20) S. Thibaut, P. Barbarat, F. Leroy, and B. A. Bernard, Human hair keratin network and curvature, Int. J. Dermatol., 46 (Suppl. 1), 7–10 (2007). (21) K. Yamauchi, A. Yamauchi, T. Kusunoki, A. Khoda, and Y. Konishi, Preparation of stable aqueous solu- tion of keratins, and physiochemical and biological properties of fi lms, J. Biomed. Mat. Res., 31, 439– 444 (1996). (22) T. Fujii and Y. Ide, Preparation of translucent and fl exible human hair protein fi lms and their properties, Biol. Pharm. Bull., 27, 1433–1436 (2004). (23) T. Tanabe, N. Okitsu, A. Tachibana, and K. Yamauchi, Preparation and characterization of keratin- chitosan composite fi lm, Biomaterials, 23, 817–825 (2002). (24) A. Yamauchi and K. Yamauchi, “Formation and Properties of Wool Keratin Films and Coatings,” in Protein-Based Films and Coatings, A. Gennadios. Ed. (CRC Press, Boca Raton, FL, 2002), pp.253–273. (25) K. Yamauchi and A. Khoda, Novel proteinous microcapsules from wool keratins, Colloids Surf., B, 9, 117–119 (1997). (26) A. Kurimoto, T. Tanabe, A. Tachibana, and K. Yamauchi, Keratin sponge: Immobilization of lysozyme, J. Biosci. Bioeng., 96, 307–309 (2003). (27) K. Katoh, T. Tanabe, and K. Yamauchi, Novel approach to fabricate keratin sponge scaffolds with con- trolled pore size and porosity, Biomaterials, 25, 4255-4262 (2004). (28) A. Tachibana, Y. Nishikawa, M. Nishino, S. Kaneko, T. Tanabe, and K. Yamauchi, Modifi ed keratin sponge: Binding of bone morphogenetic protein-1 and osteoblast differentiation, J. Biosci. Bioeng., 102, 425–429 (2006). (29) H. W. J. Harding and G. E. Rogers, ε-(γ-Glutamyl)lysine cross-linkage in citrulline-containing protein fractions from hair, Biochemistry, 10, 624–630 (1971). (30) H. Zahn, J. Föhles, M. Nienhaus, A. Schwan, and M. Spel, Wool as a biological composite structure, Ind. Eng. Chem. Prod. Res. Dev., 19, 496–501 (1980). (31) J. Menkart, L.J. Wolfram, and I. Mao, Caucasian hair, negro hair and wool: similarity and differences, J. Soc. Cosmet. Chem., 17, 769–787 (1966).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)