J. Cosmet. Sci., 71, 91–109 (March/April 2020) 91 Penetration of Surfactants into Skin LIMENG LIU , College of Pharmacy, Shenyang Pharmaceutical University, Shenyang Shi 110016, China Accepted for publication October 26, 2019. Synopsis Surfactants possess the ability to reduce surface tension at low concentrations, resulting in emulsifi cation, foaming, wetting, and solubilizing. As a versatile industrial material, surfactants can be widely used as additives in the industrial fi eld as different as textile, metal processing, mineral processing, new materials, industrial cleaning, construction, and pharmaceuticals. The most extensive application of surfactants perhaps is in the household and cosmetic industries, such as laundry detergents, dishwashing detergents, facial and body cleansers, and preparation of emulsions and creams. However, the extensive use of detergents, cleaners, and cleansers on skin may cause itching, redness, and dryness termed as surfactant-induced irritation, which is at least, partially due to surfactant penetration into skin. To understand how surfactants penetrate into skin, this review summarizes the penetration models proposed by researchers in the past two decades, including the surfactant monomer penetration model, the surfactant micelle and submicelle penetration model, and the recently proposed surfactant charge density and penetration correlation model that demonstrates the correlation between the surfactant charge density and skin penetration. INTRODUCTION Surfactants are usually constituted of hydrophilic polar groups and hydrophobic nonpolar hydrocarbon chains (or rings). Because of the special amphiphilic structure, they can reduce surface tension and facilitate foaming, emulsifying, dispersing, wetting, and osmosis, all of which contribute to their extensive applications in the chemical industry. The specifi c applications are presented in Table I. SURFACTANTS Surfactants, an abbreviation for surface active agent, are amphiphilic or amphipathic molecules which could orient themselves at the boundary of two immiscible phases. The hydrophilic or water-loving portion of the surfactants would be soluble in the aqueous solution. Conversely, the hydrophobic or water-hating portion of them would be absorbed in the nonaqueous solution (as the oleaginous phase or air) (1,2). Address all correspondence to hufanandyhu@gmail.com or hufan.gatech@edu.viax.org.
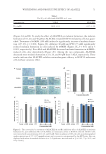
JOURNAL OF COSMETIC SCIENCE 92 On adsorption of the surfactants to the two immiscible phases, the repellent force between the phases is reduced, and, also, the free energy at the phase boundary is reduced. As more and more surfactants gather at the interface, the free energy per unit area of the interface or the surface tension gradually decreases (1). To stay steadily at the boundary of two immiscible phases, a surfactant is supposed to possess a characteristic structure, enabling itself to integrate two immiscible phases into one single phase. Generally, a surfactant monomer is mainly composed of (i) a hydrophilic part that contacts the aqueous phase in the solution and (ii) a hydrophobic part that rejects the aqueous phase (3). The hydro- philic part is usually a polar group. On the other hand, the hydrophobic part is often a nonpolar group, such as a long-chain hydrocarbon. Take water and oil, two immiscible phases, for instance the surfactants in the water and oil mixture will gather along at the water–oil interface, and the hydrophilic section of the surfactant is dissolved in the water layer, whereas the hydrophobic side is immersed in the oil layer. Only surfactant monomers are capable of reducing surface tension (1). When the concentra- tion of the surfactant increases and is saturated at the two-phase boundary, the aggrega- tion of the surfactant monomers referred to as the micelle is usually formed. Take air and liquid phases as example in the aqueous solution, when there is no space for the extra surfactant monomers at the air–liquid interface, the extra monomers will enter into the aqueous solution and their hydrophobic parts will gather together to reject the aqueous environment, forming micelles with the hydrophobic groups facing inward and the hydro- philic groups orienting outward (1). The concentration of surfactants above which micelles are formed is defi ned as the critical micelle concentration (CMC) (1,4). The diagram of the surfactant monomer and micelle, and the relationship of the surfactant monomers and micelles are described in Figures 1–3. The functions of surfactants include (i) emulsifi cation—surfactants work as emulsifi ers to reduce the water–oil interfacial tension, forming stable emulsion (5) (ii) wetting action (iii) solubilization (iv) detergency—rollup is one of the mechanisms describing how sur- factants remove sebum and dirt (6) and (v) foaming. Based on the various physicochemical properties of surfactants, they have extensive civil and industrial applications, such as laundry detergents, emulsifi ers, food additives, cosmetics, and drug delivery (5,7). This review article mainly focuses on the surfactants used in the skin-cleaning products. The classifi cation of surfactants can be based on the structure of the hydrophilic parts, con- sisting of anionic surfactants, cationic surfactants, nonionic surfactants, and amphoteric Table I Specifi c Applications of Surfactants in Different Fields Field Specifi c applications Washing industry Soap, kitchen detergent, shampoo, laundry detergent, and decolorizer Pharmaceutical industry Wetting agent, adhesive, lubricant, coating material, slow/controlled release formulation, dispersing agent, solubilizer, emulsifying agent, and germicide Food Food emulsifying agent, food defoaming agent, thickening agent, and preservative Cosmetics industry Emulsifi able paste, wetting agent, and foaming agent Petrochemical industry Lubricating oil, anticorrosive agent, and sealant, Textile industry Fluorescent whitening agent, antistatic agent, softening agent, emulsifying agent, and detergent, Agriculture Pesticide dispersing agent and wetting agent
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)